the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Unveiling the organic contribution to the initial particle growth in 3–10 nm size range
Kewei Zhang
Zhengning Xu
Fei Zhang
Zhibin Wang
Organic compounds play an important role in atmospheric particle initial growth along with sulfuric acid (SA). However, the detailed composition of newly formed particles remains limited due to analytical challenges. In this study, we conducted flow tube experiments to investigate the nanoparticle growth processes of SA and oxygenated organic molecules (OOMs, from α-pinene oxidation) system. Utilizing a custom-built scanning flow condensation particle counter (SFCPC), we report, for the first time, size-resolved measurements of the hygroscopicity parameter (κ) and organic mass fraction (forg) for particles in the 3–10 nm size range within this atmospherically relevant system. The hygroscopicity of SA decreased 49 % as particle size increased (from 0.413±0.011 at 3 nm to 0.209±0.004 at 10 nm) and declined by up to 18 % with increasing RH, which may be explained by hydration effects. In contrast, the κ values of OOMs increased with RH by as much as 57 %, potentially involving changes in oxidation product. Size-resolved forg revealed that larger particles contained a greater proportion of organics, indicating OOMs contribute more significantly to growth at larger sizes. Moreover, elevated humidity enhanced the organic contribution to particle growth by up to 81 %. Compared to 3–5 nm, this enhancement was more pronounced for 5–10 nm particles associated with the incorporation of increased yields of more volatile oxidation products and Kelvin effect. These valuable information on hygroscopicity and chemical composition of 3–10 nm particles during new particle formation and subsequent growth could further the understanding of related atmospheric mechanisms.
- Article
(1101 KB) - Full-text XML
-
Supplement
(675 KB) - BibTeX
- EndNote
Atmospheric new particle formation (NPF) is a widely observed phenomenon (Du et al., 2024; Kulmala et al., 2014) in which low-volatility gas-phase oxidation products nucleate to form aerosol particles (Lee et al., 2019). These newly formed particles can grow to become cloud condensation nuclei (CCN), potentially contributing 30 %–70 % of atmospheric CCN populations (Ren et al., 2021; Sun et al., 2024). The chemical composition of nucleating clusters and growing particles plays a fundamental role in determining both particle formation mechanisms and subsequent growth processes (Kirkby et al., 2023). However, the chemical information of the cluster and particles are still not well understood (Zhang et al., 2012; Zhao et al., 2024).
To date, the condensation of low volatility vapours, like sulfuric acid (SA) or oxidized organic compounds, is recognized as a primary mechanism driving cluster formation and particle growth (Stolzenburg et al., 2018, 2020). SA, in particular, is a key gas-phase precursor involved in the nucleation of atmospheric aerosol particles (Kulmala, 2003; Sipilä et al., 2010). Field observations (Kulmala et al., 2013; Wang et al., 2011; Yao et al., 2018; Zhang et al., 2009) and laboratory experiments (Dunne et al., 2016; Kirkby et al., 2023; Sipilä et al., 2010) demonstrate it also contributes to initial growth. But SA only contributes 10 %–50 % exceeding 3 nm (Stolzenburg et al., 2023), which indicates that SA alone rarely dominates nanoparticle growth in the atmosphere (Kuang et al., 2010). In addition, α-pinene oxidation products contribute to the growth of newly formed particles (Ehn et al., 2014; Tröstl et al., 2016). Atmospheric organic vapors play a crucial role in particle formation and growth (Kirkby et al., 2016; Riccobono et al., 2014; Zhang et al., 2004), with organic compounds potentially accounting for a substantial fraction (20 %–90 %) of submicron particle mass (Jimenez et al., 2009). And the contribution of organic species to particle growth was found to increase with particle size (Bianchi et al., 2019; Riccobono et al., 2012; Riipinen et al., 2012). However, quantitative analysis of SA or organic mass fractions in 3–10 nm particles remains challenging due to instrumental limitations in measuring chemical composition (Smith et al., 2021; Zhang et al., 2022).
Several techniques have been developed to address this measurement gap. The thermal desorption chemical ionization mass spectrometer (TDCIMS) could characterize particle composition down to 8 nm in field (Li et al., 2021), though requiring extended sampling periods (10–30 min). Keskinen et al. (2013) estimated the organic fraction in sub-2 nm using atmospheric pressure interface time-of-flight (API-TOF) mass spectrometer. To break the limitation of insufficient ion concentration, alternative approaches utilizing advanced condensation particle counters (CPCs) provided qualitative assessments of organic contributions (Kangasluoma et al., 2014; Kulmala et al., 2007; O'Dowd et al., 2002), by exploiting the inherently high number concentration of nucleation-mode particles. The nano cloud condensation nuclei counter (nano-CCNC) could further provide semi-quantitative information by applying the linear relationship between hygroscopicity parameter (κ) and organic mass fraction (forg) down to 2.5 nm (Wang et al., 2015). Though its stabilization period (∼2 min for a single supersaturation) limits applicability to rapidly growing newly formed particles (e.g., 35.7 nm h−1 in urban Shanghai, Xiao et al., 2015). More recently, the scanning flow condensation particle counter (SFCPC) has demonstrated capability for the κ–forg relationship by achieving supersaturation adjustments within ∼2 s through flow rate modulation (Zhang et al., 2023), making it particularly suitable for organic fraction determination in 3–10 nm.
In this study, we conducted a series of laboratory nucleation and growth experiments using a custom-built flow tube reactor. SO2 and α-pinene were employed as gas-phase precursors to generate SA and organics (OOMs, oxygenated organic molecules), respectively. Experiments were performed under purely inorganic, purely organic, and mixed precursor conditions with varying ratios, across a wide RH range (20 %–80 %). We first measured the κ values of 3–10 nm particles formed from the oxidation products through SFCPC. Then our analysis established size-resolved linear relationships between κ and forg for SA-OOMs mixed particles, enabling quantitative determination of organic contributions. Furthermore, we systematically examined the effects of particle size, gas precursor concentration ratio, and humidity on both particle hygroscopicity and organic contribution. Based on these experimental results, this study aims to quantitatively investigate the distinct roles of sulfuric acid and oxygenated organics during nanoparticle growth, and to clarify how environmental conditions modulate the chemical composition and water uptake of sub-10 nm particles.
2.1 Experimental set-up
A custom-built flow tube reactor was used to perform a series of laboratory studies on nucleation and growth. This flow tube consisted of a 25 cm long×1 cm i.d. quartz tube (19.6 mL in volume) fitted with stainless steel adapter on each end. The entrance was coupled with two union cross in line to introduce gas precursors. As shown in Fig. 1, the water vapor generated by passing N2 through water and O3 from a UVP ozone generator (model SOG-2, Analytik Jena US) were introduced into the main gas flow in the first union cross. The SA and OOMs in all experiments were generated from SO2 and α-pinene. These precurses were diluted from a gas cylinder containing 0.6 % SO2 and obtained from a gas cylinder with 50 ppmv α-pinene, respectively. The carrier flow, water vapor, O3, SO2 and α-pinene were mixed in the second union cross and then introduced into flow tube reactor. To initiate photolysis reactions in the system, a UV lamp (model 11SC-1, Analytik Jena US) with a length of 5.38 cm was installed at the entrance of the quartz tube, emitting ultraviolet light at a wavelength of 254 nm. At the exist of flow tube reactor, the temperature and RH were measured by a humidity sensor (model SHT85, Sensirion) with precision of ±1.5 %. The O3 concentration was monitored by an ozone analyser (model 49i, Thermo Fisher Scientific). The concentrations of other gaseous precursors were derived from their mixing ratios, and the molecular composition of the flow tube products was not directly measured in this study.
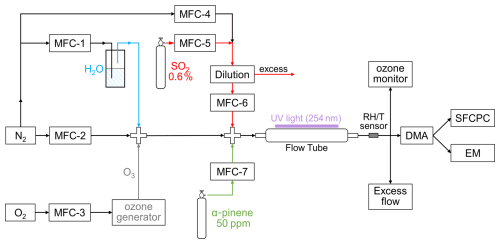
Figure 1Schematic of the flow tube reactor and experimental setup. Precursers (SO2, α-pinene, O3 and H2O) were introduced into the flow tube at a total flow rate of . SO2 and α-pinene were subsequently oxidized by O3 under UV irradiation (254 nm). The resulting oxidation products were classified using a nano-DMA, with particle hygroscopicity and organic content characterized by SFCPC and EM.
SA and OOMs was used in this work to represent inorganic and organic components in atmosphere respectively. For pure inorganic experimental groups, H2SO4 was generated in situ via the reaction of OH radicals with SO2 in the presence of water vapor. When the SO2, O3 and H2O were mixed and introduced into the flow tube reactor, O3 underwent photolysis to produce O(1D) atoms, which then react with water vapor to generate OH radicals. These OH radicals interact with SO2, forming the HOSO2 adduct, which subsequently decomposed to produce SO3 and HO2 (Jayne et al., 1997; Lovejoy et al., 1996). In the reaction of SO3 with water vapor, two H2O molecules or one H2O dimer per SO3 molecule were required, ultimately resulting in the formation of H2SO4 (Berndt et al., 2005). For pure organic experimental groups, OOMs were produced from organic peroxides formed from oxidation reactions of α-pinene (Kirkby et al., 2016; Lee et al., 2019). α-pinene was exposed to ozone and also to hydroxyl radicals (OH) due to the unavoidable production of OH from ozone photolysis and secondary reactions. Although detailed molecular composition information could not obtained in our work, the ozonolysis pathway is generally understood to proceed via Criegee intermediates, leading to various peroxy radicals and subsequent low-volatility products (Iyer et al., 2021; Yang et al., 2025). Similarly, OH-initiated oxidation proceeds mainly via OH addition, forming peroxy radicals that further react to produce condensable organic species (Berndt et al., 2016; Kang et al., 2025). While the present study did not estimate the OH concentration and further elucidate specific mechanistic pathways, this simplification is justified because, to the best of our knowledge, no existing studies have clearly demonstrated significant differences in the hygroscopic performance of pure OOMs derived from different α-pinene oxidation pathways.
The SO2 and α-pinene concentration in experiments were regulated by setting the mixing ratio of SO2 flow rate and α-pinene flow rate to the total flow rate. The total flow rate was 3000 mL min−1, and the residence time in flow tube reactor was about 0.4 s. The all flow rates in this flow tube reactor were set by mass flow controllers (MFCs; MFC. 1–4, model GT130D, Gas Tool Instruments Co., Ltd.; MFC. 5–7, model Sevenstar CS200, NAURA Technology Group Co., Ltd.). The conditions of 24 experimental groups conducted to research the organic contribution to new formed particles in the initial growth stage were summarized in Table S1 in the Supplement. To research the RH impacts to the initial growth process, the experiments were divided to four series conducted under RH 20 %, 40 %, 60 % and 80 %. For pure inorganic (Exp. A) and mixture groups (Exp. C–F), the concentration of SO2 was set as a constant value and the concentration of α-pinene was regulated based on the (concentration ratio of gas precursors α-pinene and SO2, 0.1–1). The pure organic experiments (Exp. B) were conducted with much higher concentration than that in mixture experimental groups to generate sufficient 3–10 nm particles (number concentration larger than 1000 # per cm3). The temperature in all experiments was around 28 °C when the reaction was stable. O3 concentration obtained by Ozone monitor was ∼200 ppb with a regulable O2 flow rate.
2.2 Determination of κ
The hygroscopicity of nanoparticles was measured with a custom-designed SFCPC. The setup of SFCPC system has been described previously (Zhang et al., 2023), and only a brief summary is presented here. The aerosol particles were passed through a neutralizer (X-ray, TSI model 3080), and a nano-differential mobility analyzer (nano-DMA, TSI model 3086) was used to select charged monodisperse particles in diameter range of 3–10 nm (with intervals of 0.2 nm in the 3–4 nm range and intervals of 0.5 nm in the 4–10 nm range). Considering the negligible probability of double-charge for particles below 20 nm (Fuchs, 1963; Wiedensohler et al., 1986; Wiedensohler and Fissan, 1988), no double-charge correction was applied in the studied size range. SFCPC, which was improved from a water-based condensation particle counter (WCPC, TSI model 3788) and could change the supersaturation fast by altering the sample flow, was deployed to count the activated particle concentration (N). And an electrometer (EM, TSI model 3068B) was operated in parallel to measure the total particle concentration (Ntotal), where the counting efficiency of sampled particles (fcount) in different supersaturation (S) conditions could be obtained by . S distributions of SFCPC were calibrated by tungsten oxides (WOx) particles generated from a WOx generator (Model 7.860, Grimm Aerosol Technik; Steiner, 2011), and twelve S conditions were setup to meet the requirements of κ value measurements. The effective S ranged from 7.7 % to 73.1 %, a sufficiently broad range to activate the SA and OOMs corresponding fcount in 20 %–80 %. For the measurement part, particles from flow tube reactor were selected with discrete diameters and the S could be calculated through the measured fcount. Then the κ values could be obtained from dry diameter (Dd) and its corresponding S based on κ-Köhler equation (Petters and Kreidenweis, 2007). The equation was applied following Eq. (2) in Zhang et al. (2023), in which the surface tension of water (0.072 N m−1) was used.
2.3 Determination of forg
The linear relationship between chemical composition and hygroscopicity of CCN sized particles has been found both in laboratory experiments and field measurements (Dusek et al., 2010; Pöhlker et al., 2023; Vogel et al., 2016; Zhou et al., 2024). Our previous work extended the linear relationship between κ and forg to 3–10 nm size range for AS and levoglucosan/sucrose mixed particles, which were generated via Electrospray from solutions with known mixing ratios (Wang et al., 2015; Zhang et al., 2023). Zhang et al. (2025) assessed the uncertainty arising from the dependence of κ on particle size, also based on Electrospray-generated particles (AS and cis-pinonic acid). In this work, we introduced κ–forg linear relationship into the mixing products from flow tube – a setup designed to simulate atmospheric processes – in order to explore the organic content in the particulate phase. Furthermore, we established size-resolved κ–forg linear relationship to eliminate the mentioned uncertainty. The application of the κ–forg linear relationship relies on the assumption of ideal internal mixing within the particles. Under our experimental conditions, for in situ freshly formed 3–10 nm particles, the characteristic mixing times are short, and organic-inorganic mixtures are likely to remain liquid and well-mixed (Cheng et al., 2015). Therefore, the ideal internal mixing assumption is reasonable. For SA-OOMs mixture, we use the κ values of pure organic (OOMs) and inorganic (SA) experiment groups to represent the hygroscopicity of the organic and inorganic component in the mixture particles, respectively. Although the organic and inorganic components in mixture may not be identical to those in pure organic and inorganic particles of the same size under the same RH due to the potential change of oxidation processes, this simplification is a necessary given the current inability to directly measure the composition and component-specific hygroscopicity of particles in the 3–10 nm size range. To further reduce uncertainties associated with the linear relationship, the κ values of pure organic and inorganic particles were taken from the fitted lines to serve as a reference baseline, as shown in Fig. S1 in the Supplement. Based on the size-resolved κ–forg linear relationship (colored solid lines) and the measured κ values (grey dashed line) of the SA-OOMs mixture, the corresponding forg values (colored dashed lines) for particles generated in the flow tube reactor were derived using Eq. (1):
where forg and κorg is the mass fraction and hygroscopicity of pure organics (OOMs in this work), respectively; κinorg is the hygroscopicity of pure inorganic component (SA in this work); κ is the hygroscopicity of the measured particles nucleation and growth process in flow tube reactor.
3.1 Hygroscopicity
The results of pure inorganic groups and organic groups were shown in Fig. 2. The hygroscopicity of 3–10 nm SA particles formed by the oxidation products of SO2 have significant size dependence. κ values would decrease with Dd, from 0.413±0.011 for 3 nm to 0.209±0.004 for 10 nm under RH=20 %. It should be noted that the quartz tube was replaced for every single experiment group to eliminate the potential contamination between experiments. Moreover, prior to the hygroscopicity measurements, the oxidation products of SO2 were characterized using an iodide chemical ionization mass spectrometry (Vocus AIM), which detected no significant organic compound signals in the gas phase (Fig. S2 in the Supplement). Therefore, this decreasing trend cannot be attributed to organic contamination. Considering the sulfuric acid and water binary nucleation is the basic mechanism for the SA particle formation, the water molecules plays a significant role through hydration (Kulmala et al., 1998; Lee et al., 2019; Stolzenburg et al., 2023; Yu et al., 2017). The initial formation of H2SO4•H2O molecular clusters is followed by rapid addition of further H2O molecules and these stepwise hydrates process ultimately leads to particles covered with water during growth (Couling et al., 2003; Matsubara et al., 2009). The measured particles were thought as (H2SO4)m-(H2O)n rather than pure H2SO4. Consequently, the decreasing trend of κ with particle size may be explained by the increasing water content, which lowers the particles' water uptake capacity relative to their dry mass.
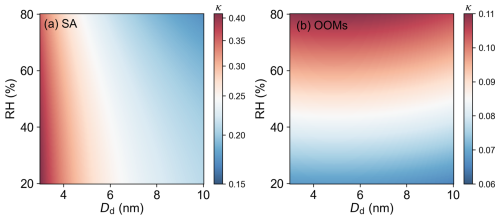
Figure 2Dependence of hygroscopicity on particle size and RH: (a) SA formed from SO2, and (b) OOMs from α-pinene oxidation. Color scale represents the κ values derived from forg measurements using SFCPC and EM, where the values were fitted from measurement data to illustrate κ variation trend more clearly.
Hygroscopicity of SA particles also revealed a consistent decrease with rising RH: at RH=80 %, κ values declined to 0.361±0.013 for 3 nm and 0.171±0.011 for 10 nm particles, decreasing 13 % and 18 % compared to RH=20 %. This demonstrates that SA particles exhibit reduced hygroscopicity under higher humidity conditions. Although bases such as ammonia/amine could in principle suppress the hygroscopicity of SA particles (Yishake et al., 2025), the κ of an SA-ammonia/amine mixture would be expected to increase with RH, because a higher SA fraction (more acidic) is expected at higher RH (Chen et al., 2018). This κ–RH dependence is opposite to the trend observed in our study. Moreover, there was no identified source of bases in our system, so ammonia contamination can largely be ruled out. Furthermore, previous studies have shown that the average number of water molecules hydrating each H2SO4 molecule increases with RH (Kurtén et al., 2007; Temelso et al., 2012; Zollner et al., 2012). Therefore, the observed decrease in hygroscopicity can be attributed to the particles already containing more water at higher RH, resulting in a reduced capacity for additional water uptake.
The κ values reported in previous studies are summarized in Table 1. The measured hygroscopicity results in this work are much lower than those reported in previous studies (0.68–0.9, Petters and Kreidenweis, 2007; Shantz et al., 2008; Sullivan et al., 2010), which predicted by thermodynamic model in 30–80 nm based on the model parameters provided by Clegg et al. (1998). The hygroscopicity of newly formed sulfuric acid nanoparticles in CLOUD chamber was examined by a nano hygroscopicity tandem differential mobility analyser (nano-HTDMA) and the reported κ values were 0.64±0.02 and 0.52±0.02 for 10 and 15 nm, respectively (Kim et al., 2016). The reported variation trend in particle size aligns with our findings but the κ values were much larger than that in this work. While both Kim et al. (2016) and our study report κ values of the oxidation products of SO2, the measurement methods used are fundamentally different. Kim et al. (2016) employed a nano-HTDMA, which measures hygroscopic growth under subsaturated conditions. In contrast, our SFCPC method derives κ values by activating particles under supersaturated conditions. As discussed in previous studies, the measurement condition can induce inherently different κ values (Biskos et al., 2009; Massling et al., 2023). Specifically, SA particles are expected to exhibit comparably higher hygroscopic growth under subsaturated conditions and lower CCN activity at supersaturation. Therefore, this methodological difference is likely the primary factor contributing to this discrepancy in κ values. In addition, for sub-10 nm particles, the enhanced Kelvin effect (compared to that for larger CCN-size particles) dramatically raises the energy barrier for vapor condensation, directly contributing to suppressed hygroscopicity. Furthermore, the potential size-dependent influence on phase state may reflect a tendency for nanoparticles to remain in a liquid or mixed phase (Cheng et al., 2015). These nano-size effects on the thermodynamic and physical properties of aerosol particles are likely significant contributing factors in explaining the distinctively lower κ values observed in our study. As far as we know, direct measurements of the κ of sulfuric acid particles remain limited in the literature, and our results is the first measurement under supersaturation condition in 3–10 nm range.
Table 1Summary of κ values of SA and oxidation products of α-pinene reported in previous studies and this work.
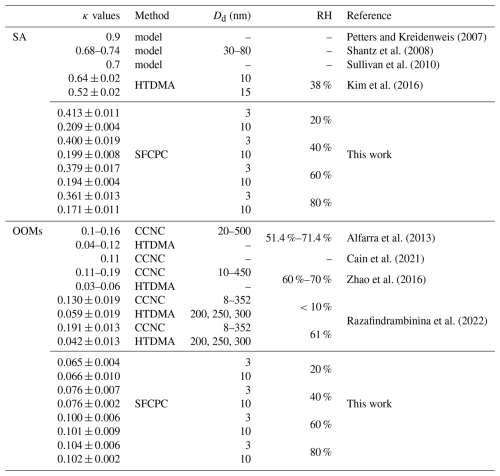
Different from the SA results, the OOMs particles formed by the oxidation products of α-pinene have almost constant κ values in the 3–10 nm diameter range. As shown in Fig. 2b, κ of the pure organic group under 20 % RH is ∼0.065. This result indicates that the OOMs particles exhibit non-hygroscopicity compared to SA particles and its hygroscopicity has no significant dependence on particle diameter. While the detailed chemical composition of OOMs in these nanoparticles may vary with size due to volatility-dependent partitioning and the Kelvin effect, the observed constancy in κ suggests that the effective hygroscopic properties of the condensing mixture do not change significantly within this size range. This finding is consistent with Frosch et al. (2011), who also observed almost constant κ values (0.11±0.02) for 50–150 nm particles from α-pinene oxidation. However, to our knowledge, research on particle size dependence is very rare, and no studies have specifically examined the hygroscopicity of α-pinene oxidation particles in the 3–10 nm size range. Alfarra et al. (2013) measured the hygroscopicity of particles formed from α-pinene oxidation via both OH radicals and O3 using CCNC. Our results align closely with their reported κ values (0.1–0.16), which were also observed under supersaturation conditions. Furthermore, numerous studies have investigated the hygroscopicity of α-pinene oxidation products using CCNC or HTDMA, reporting a wide range of κ values (0.03–0.19, Cain et al., 2021; Duplissy et al., 2011; Engelhart et al., 2008; Massoli et al., 2010; Razafindrambinina et al., 2022; Wang et al., 2019; Zhao et al., 2016). All experimental results reported here fall within the established range of values found in existing publications.
Contrasting with the negligible size dependence, the measured κ values of OOMs exhibited a pronounced increase with RH, rising by 57 % from 20 % to 80 % RH. This finding aligns with Razafindrambinina et al. (2022), who reported higher κ values for laboratory-generated α-pinene oxidation products under humid conditions (κ=0.191 at 75 %–80 % RH) compared to dry conditions (κ=0.130 at RH<10 %). Similarly, Luo et al. (2024) observed that the molecular composition of α-pinene oxidation products evolves with increasing RH. While direct molecular-level speciation from our measurements is unavailable, previous studies on α-pinene oxidation systems provide an explanatory framework. The work of Yuan et al. (2017) suggests that in the presence of water vapor, particles formation may promote the generation of more stable Criegee intermediates, leading to the production of more hygroscopic materials in monoterpene systems. This is supported by evidence of increased formation of oxygenated functional groups, such as multifunctional carboxylic acids, under humid conditions (Poulain et al., 2010). The hygroscopicity of such compounds is intrinsically higher, as the κ increases with the functionality in the following order: (-CH3 or -NH2) < (-OH) < (-COOH or C=C or C=O) (Han et al., 2022). Consequently, the observed increase in κ values at higher RH in this study is attributed to the likely formation of more hygroscopic components.
3.2 Organic mass fraction
The forg values of 3–10 nm particles was determined using the κ–forg linear relationship as described in Sect. 2.3. Figure 3 presents the retrieved forg results for four experimental groups with varying ratios under 20 % RH. For 3–10 nm SA-OOMs mixed particles, the median mass fraction of OOMs increased from 7.9 % to 36.9 % as the ratio increased from 0.1 to 1. Considering that ozone was always excessive in the flow tube reactor, the oxidation products of α-pinene are expected to increase with rising precursor concentration. Consequently, the organic content in the particle phase should be proportional to the concentration of condensable OOMs in the gas phase. This significant increasing trend has also been reported in previous studies. Li et al. (2022) observed that the ratio of particulate organics to sulphate in urban field measurements was positively correlated with the ratio of gaseous condensable organic oxidation products to sulfuric acid. Comprehensive modelling study demonstrated that terpene-rich air masses containing abundant low volatility oxidation products substantially enhanced the condensational growth of nano-particles and dominated the initial growth stage in sub-10 nm, with contribution as high as 95 % (Huang et al., 2016). Our experimental results align with these findings, demonstrating that increased organic precursor concentrations significantly elevates the contribution of OOMs to the growth of 3–10 nm particles.

Figure 3Organic mass fraction as a function of the precursor concentration ratio for SA-OOMs mixture particles at RH 20 %. The box plots display the interquartile range (IQR), with the central line denoting the median and whiskers extending to 1.5×IQR. The coloured scatter points represent the mean forg values for each selected Dd, as indicated by the colour scale.
At the same time, our results reveal that the organic content of large particles is generally higher than that of smaller particles (coloured scatter points in Fig. 3). To further investigate the size-dependent effect on the chemical composition of nano-particles, the experimental data were analysed by categorizing particles into three distinct size ranges (3–5, 5–7, and 7–10 nm). Figure 4 shows the results from Exp. C and Exp. F, which had the lowest and highest ratios of 0.1 and 1, respectively. The forg values for 3–5, 5–7, and 7–10 nm SA-OOMs particles in Exp. C are 5.7 %, 9.8 % and 11.3 %, respectively. Similarly, particles formed in Exp. F contained 27.5 %, 39.5 %, and 43.2 % organics, respectively. These results indicate that the forg exhibited a consistent increase with Dd across the range of the precursors mixing ratios examined in this work. Furthermore, these findings demonstrate that α-pinene oxidation products contributed progressively more to particulate phase in the SA-OOMs system as particle size increased.
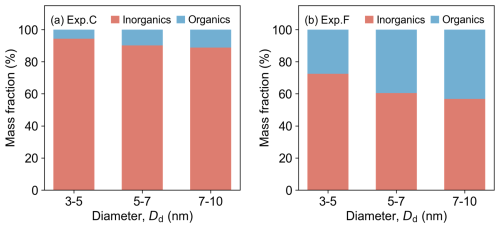
Figure 4Organic mass fraction of SA-OOMs mixtures as a function of particles diameter range with two precursors mixing ratios: (a) 0.1 for Exp. C, (b) 1 for Exp. F. Red and blue bars represent forg values of inorganics (SA) and organics (OOMs). Note that the size ranges are defined as semi-open intervals, except for the first bin which includes its lower bound.
Several studies have also investigated the chemical composition of newly formed particles in the initial growth stage. Kim et al. (2016) employed nano-HTDMA measurements in the CLOUD chamber to determine κ values, from which volume fractions were derived through linear relationships. Their work revealed that the volume fraction of dimethylamine sulphate in sulfuric acid-dimethylamine systems increased substantially from 0.20–0.29 for 10 nm particles to 0.58–0.92 at 15 nm. Similarly, Keskinen et al. (2013) observed a progressive enhancement in organic volume fraction from 0.4 at 2 nm to 0.9 at 63 nm, while noting the existing measurement challenges for particles between 2–15 nm where chemical characterization remains particularly difficult. More recently, Li et al. (2022) achieved direct measurements of size-resolved molecular composition using TDCIMS, demonstrating a clear increase in organic mass fraction with particle diameter across the 8–40 nm range in urban Beijing. Other studies have also indicated that particle growth mechanisms exhibit a dependence on particle size (Riipinen et al., 2012). The contribution of SA decreased as particle size increased (Xiao et al., 2015), while organic compounds increasingly dominated the growth process, showing a strong size-dependent effect (Riccobono et al., 2012). Our experimental results align well with these previous studies, confirming that organic contributions to nanoparticle growth exhibit consistent size dependence, while additionally providing novel composition data for freshly nucleated particles as small as 3 nm, thereby extending the current understanding of early particle growth.
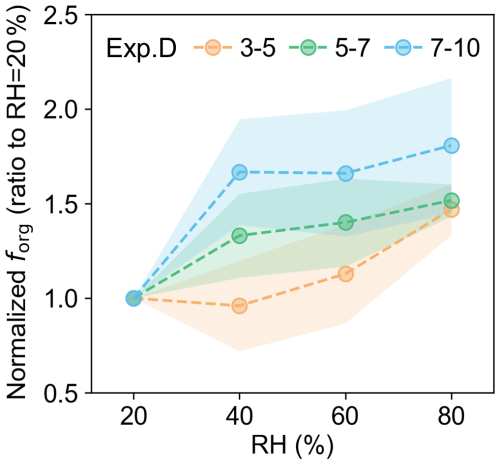
3.3 Effect of relative humidity
To investigate the effect of humidity on the initial growth of newly formed particles, we increased RH from 20 % to 80 % and measured the organic content across this range. The retrieved forg values were similarly grouped by particle size, following the same methodology as in the size-dependence analysis. The results from all RH experimental groups (20 %–80 %) were compared with those under 20 % RH to examine the relative change in organic contribution with increasing RH (Fig. 5). The enhancement of organic contribution to particle growth by humidity exhibits a marked difference between particles below and above 5 nm. Taking Exp. D as an example, the forg for 3–5 nm particles began to increase from 60 % RH, with a relative increment reaching approximately 47 % at 80 % RH. For particles in the 5–7 and 7–10 nm size ranges, forg exhibited a sharp increase starting at 40 % RH and remained almost stable within the 40 %–80 % RH range. Compared to the condition at 20 % RH, the organic contribution to particle growth was enhanced by about 52 % and 81 %, respectively. We speculate that these behaviours arise from the competing influences of humidity on the physicochemical properties of α-pinene oxidation products and the Kelvin effect. For such small nanoparticles, the partitioning of a molecule into the particulate phase is influenced by both its volatility and the Kelvin effect (Riipinen et al., 2012). Previous molecular measurements in both gas and particle phases have reported increased yields at elevated RH (Poulain et al., 2010). Concurrently, Surdu et al. (2023) observed that α-pinene oxidation products become more volatile under humid conditions. The relative stability of forg in the 3–5 nm particles at low RH condition may thus be explained by a balance between these two competing mechanisms, where the heightened Kelvin effect presents a significant barrier to condensation. For larger particles, the diminished Kelvin effect facilitates the condensation of organic compounds, allowing even more volatile products to contribute to nanoparticle growth. The distinct response patterns, where the enhancement occurred gradually for 3–5 nm particles but sharply for larger particles, suggest that the Kelvin effect plays a more dominant role for the smallest particle growth at lower RH. Overall, increased RH enhances the organic contribution by altering the properties of α-pinene oxidation products, with a more pronounced effect observed for larger particles.
In this study, a flow tube reactor was employed to conduct a series of experiments investigating nucleation and particle growth across a wide range of RH (20 %–80 %). SO2 and α-pinene served as gas-phase precursors for the formation of SA and OOMs in all experiments, with the precursor concentration ratio varying from 0.1 to 1. A custom-designed SFCPC was utilized to quantify the size-resolved hygroscopicity. Based on the linear relationship between κ and forg, the organic mass fraction of freshly formed particles was determined within the 3–10 nm diameter range.
Our work provides quantitative insight into the distinct hygroscopic behaviours of SA and OOMs. For SA, κ exhibited significant size dependence, decreasing from 0.413±0.011 at 3 nm to 0.209±0.004 at 10 nm under 20 % RH. When RH was elevated to 80 %, κ further declined by 18 % (to 0.171±0.011 at 10 nm). This decrease may be explained by the formation of (H2SO4)m-(H2O)n complexes. The hydration process eventually results in water-covered particles, reducing their capacity for further water uptake. The measured κ values in this work are much lower than those reported in previous studies and this discrepancy may be attributed to the nano size effect and different measurement techniques. In contrast, κ of OOMs remained relatively constant across the 3–10 nm size range but increased with rising RH (from 0.069±0.003 to 0.108±0.004 as RH rose from 20 % to 80 %) potentially involving changes in the physicochemical properties of the oxidation products. These experimental findings are consistent with previously reported κ values for α-pinene oxidation particles, which typically range from 0.03 to 0.19. The derived organic content of SA-OOMs mixed particles consistently increased with higher ratios, indicating enhanced contribution of organics to the particulate phase. Furthermore, forg exhibited a consistent increase with particle size. Specifically, forg values increased from 5.67 % to 11.25 % and from 27.47 % to 43.22 % for of 0.1 and 1, respectively. This demonstrated that the contribution of α-pinene oxidation products to particulate phase in the SA-OOMs system became more pronounced at larger particles. Our results align with previous studies, further confirming the size dependence of organic contributions to nanoparticle growth and additionally providing novel composition data down to 3 nm. The effects of RH on chemical composition revealed distinct trends in forg across different size ranges: for 3–5 nm particles, forg varied negligibly until 60 % RH, while for 5–10 nm particles, it increased with RH and had nearly stabilized at 40 % RH. These observations may be associated with a combination of factors including the Kelvin effect as well as increased volatility and higher yields under elevated RH.
To the best of our knowledge, this is the first study to measure the hygroscopicity (κ) of particles composed of SA and α-pinene-derived OOMs down to 3 nm. Given that direct measurements of chemical composition for newly formed 3–10 nm particles remain limited in the literature, our study provides important, quantitative, and size-resolved organic content data in this nano size range. The experimental results of SA-OOMs mixture indicate that OOMs contribute significantly to the particulate phase, with their mass fraction increasing with particle size, ratio, and RH. These findings provide valuable supplementary information for advancing our understanding of new particle formation and subsequent growth. Looking ahead, exploring of multi-precursor systems, longer oxidation times for better simulating aging processes, and the further deployment of the SFCPC in field measurements will yield deeper insights into the chemical composition of atmospheric aerosols.
Data are available upon request to the corresponding author.
The supplement related to this article is available online at https://doi.org/10.5194/acp-26-2241-2026-supplement.
KZ and ZW contributed to the methodology, data curation, and writing of the original draft. ZX and FZ contributed to the reviewing and editing. ZW contributed to the supervision, funding acquisition, conceptualization, investigation, data curation, writing, reviewing, and editing.
At least one of the (co-)authors is a member of the editorial board of Atmospheric Chemistry and Physics. The peer-review process was guided by an independent editor, and the authors also have no other competing interests to declare.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. The authors bear the ultimate responsibility for providing appropriate place names. Views expressed in the text are those of the authors and do not necessarily reflect the views of the publisher.
This research has been supported by the National Natural Science Foundation of China (grant nos. 41805100, 91844301, and 42005086) and the National Key Research and Development Program of China (grant no. 2022YFC3703505).
This paper was edited by Chiara Giorio and reviewed by four anonymous referees.
Alfarra, M. R., Good, N., Wyche, K. P., Hamilton, J. F., Monks, P. S., Lewis, A. C., and McFiggans, G.: Water uptake is independent of the inferred composition of secondary aerosols derived from multiple biogenic VOCs, Atmos. Chem. Phys., 13, 11769–11789, https://doi.org/10.5194/acp-13-11769-2013, 2013.
Berndt, T., Böge, O., Stratmann, F., Heintzenberg, J., and Kulmala, M.: Rapid formation of sulfuric acid particles at near-atmospheric conditions, Science, 307, 698–700, https://doi.org/10.1126/science.1104054, 2005.
Berndt, T., Richters, S., Jokinen, T., Hyttinen, N., Kurtén, T., Otkjær, R. V., Kjaergaard, H. G., Stratmann, F., Herrmann, H., Sipilä, M., Kulmala, M., and Ehn, M.: Hydroxyl radical-induced formation of highly oxidized organic compounds, Nat. Commun., 7, 13677, https://doi.org/10.1038/ncomms13677, 2016.
Bianchi, F., Kurtén, T., Riva, M., Mohr, C., Rissanen, M. P., Roldin, P., Berndt, T., Crounse, J. D., Wennberg, P. O., Mentel, T. F., Wildt, J., Junninen, H., Jokinen, T., Kulmala, M., Worsnop, D. R., Thornton, J. A., Donahue, N., Kjaergaard, H. G., and Ehn, M.: Highly oxygenated organic molecules (HOM) from gas-phase autoxidation involving peroxy radicals: A key contributor to atmospheric aerosol, Chem. Rev., 119, 3472–3509, https://doi.org/10.1021/acs.chemrev.8b00395, 2019.
Biskos, G., Buseck, P. R., and Martin, S. T.: Hygroscopic growth of nucleation-mode acidic sulfate particles, J. Aerosol Sci., 40, 338–347, https://doi.org/10.1016/j.jaerosci.2008.12.003, 2009.
Cain, K. P., Liangou, A., Davidson, M. L., and Pandis, S. N.: α-pinene, limonene, and cyclohexene secondary organic aerosol hygroscopicity and oxidation level as a function of volatility, Aerosol Air Qual. Res., 21, 200511, https://doi.org/10.4209/aaqr.2020.08.0511, 2021.
Chen, H., Chee, S., Lawler, M. J., Barsanti, K. C., Wong, B. M., and Smith, J. N.: Size resolved chemical composition of nanoparticles from reactions of sulfuric acid with ammonia and dimethylamine, Aerosol Sci. Technol., 52, 1120–1133, https://doi.org/10.1080/02786826.2018.1490005, 2018.
Cheng, Y., Su, H., Koop, T., Mikhailov, E., and Pöschl, U.: Size dependence of phase transitions in aerosol nanoparticles, Nat. Commun., 6, 5923, https://doi.org/10.1038/ncomms6923, 2015.
Clegg, S. L., Brimblecombe, P., and Wexler, A. S.: Thermodynamic Model of the System H+--Na+---Cl−-H2O at 298.15 K, J. Phys. Chem. A, 102, 2155–2171, https://doi.org/10.1021/jp973043j, 1998.
Couling, S. B., Fletcher, J., Horn, A. B., Newnham, D. A., McPheat, R. A., and Gary Williams, R.: First detection of molecular hydrate complexes in sulfuric acid aerosols, Phys. Chem. Chem. Phys., 5, 4108, https://doi.org/10.1039/B306620G, 2003.
Du, W., Zhao, J., Dada, L., Xu, W., Wang, Y., Shi, Y., Chen, X., Kokkonen, T. V., Cai, J., Zhang, Y., Wang, Q., Cai, R., Zha, Q., Zhou, L., Li, Z., Yu, F., Fu, P., Hu, F., Wang, Z., Worsnop, D. R., Bianchi, F., Kerminen, V.-M., Kulmala, M., and Sun, Y.: Impacts of enhanced new-particle growth events above urban roughness sublayer on cloud condensation nuclei, One Earth, 101169, https://doi.org/10.1016/j.oneear.2024.12.005, 2024.
Dunne, E. M., Gordon, H., Kürten, A., Almeida, J., Duplissy, J., Williamson, C., Ortega, I. K., Pringle, K. J., Adamov, A., Baltensperger, U., Barmet, P., Benduhn, F., Bianchi, F., Breitenlechner, M., Clarke, A., Curtius, J., Dommen, J., Donahue, N. M., Ehrhart, S., Flagan, R. C., Franchin, A., Guida, R., Hakala, J., Hansel, A., Heinritzi, M., Jokinen, T., Kangasluoma, J., Kirkby, J., Kulmala, M., Kupc, A., Lawler, M. J., Lehtipalo, K., Makhmutov, V., Mann, G., Mathot, S., Merikanto, J., Miettinen, P., Nenes, A., Onnela, A., Rap, A., Reddington, C. L. S., Riccobono, F., Richards, N. A. D., Rissanen, M. P., Rondo, L., Sarnela, N., Schobesberger, S., Sengupta, K., Simon, M., Sipilä, M., Smith, J. N., Stozkhov, Y., Tomé, A., Tröstl, J., Wagner, P. E., Wimmer, D., Winkler, P. M., Worsnop, D. R., and Carslaw, K. S.: Global atmospheric particle formation from CERN CLOUD measurements, Science, 354, 1119–1124, https://doi.org/10.1126/science.aaf2649, 2016.
Duplissy, J., DeCarlo, P. F., Dommen, J., Alfarra, M. R., Metzger, A., Barmpadimos, I., Prevot, A. S. H., Weingartner, E., Tritscher, T., Gysel, M., Aiken, A. C., Jimenez, J. L., Canagaratna, M. R., Worsnop, D. R., Collins, D. R., Tomlinson, J., and Baltensperger, U.: Relating hygroscopicity and composition of organic aerosol particulate matter, Atmos. Chem. Phys., 11, 1155–1165, https://doi.org/10.5194/acp-11-1155-2011, 2011.
Dusek, U., Frank, G. P., Curtius, J., Drewnick, F., Schneider, J., Kürten, A., Rose, D., Andreae, M. O., Borrmann, S., and Pöschl, U.: Enhanced organic mass fraction and decreased hygroscopicity of cloud condensation nuclei (CCN) during new particle formation events, Geophys. Res. Lett., 37, 2009GL040930, https://doi.org/10.1029/2009GL040930, 2010.
Ehn, M., Thornton, J. A., Kleist, E., Sipilä, M., Junninen, H., Pullinen, I., Springer, M., Rubach, F., Tillmann, R., Lee, B., Lopez-Hilfiker, F., Andres, S., Acir, I.-H., Rissanen, M., Jokinen, T., Schobesberger, S., Kangasluoma, J., Kontkanen, J., Nieminen, T., Kurtén, T., Nielsen, L. B., Jørgensen, S., Kjaergaard, H. G., Canagaratna, M., Maso, M. D., Berndt, T., Petäjä, T., Wahner, A., Kerminen, V.-M., Kulmala, M., Worsnop, D. R., Wildt, J., and Mentel, T. F.: A large source of low-volatility secondary organic aerosol, Nature, 506, 476–479, https://doi.org/10.1038/nature13032, 2014.
Engelhart, G. J., Asa-Awuku, A., Nenes, A., and Pandis, S. N.: CCN activity and droplet growth kinetics of fresh and aged monoterpene secondary organic aerosol, Atmos. Chem. Phys., 8, 3937–3949, https://doi.org/10.5194/acp-8-3937-2008, 2008.
Frosch, M., Bilde, M., DeCarlo, P. F., Jurányi, Z., Tritscher, T., Dommen, J., Donahue, N. M., Gysel, M., Weingartner, E., and Baltensperger, U.: Relating cloud condensation nuclei activity and oxidation level of α-pinene secondary organic aerosols, J. Geophys. Res., 116, D22212, https://doi.org/10.1029/2011JD016401, 2011.
Fuchs, N. A.: On the stationary charge distribution on aerosol particles in a bipolar ionic atmosphere, Geofis. Pura Appl., 56, 185–193, https://doi.org/10.1007/BF01993343, 1963.
Han, S., Hong, J., Luo, Q., Xu, H., Tan, H., Wang, Q., Tao, J., Zhou, Y., Peng, L., He, Y., Shi, J., Ma, N., Cheng, Y., and Su, H.: Hygroscopicity of organic compounds as a function of organic functionality, water solubility, molecular weight, and oxidation level, Atmos. Chem. Phys., 22, 3985–4004, https://doi.org/10.5194/acp-22-3985-2022, 2022.
Huang, X., Zhou, L., Ding, A., Qi, X., Nie, W., Wang, M., Chi, X., Petäjä, T., Kerminen, V.-M., Roldin, P., Rusanen, A., Kulmala, M., and Boy, M.: Comprehensive modelling study on observed new particle formation at the SORPES station in Nanjing, China, Atmos. Chem. Phys., 16, 2477–2492, https://doi.org/10.5194/acp-16-2477-2016, 2016.
Iyer, S., Rissanen, M. P., Valiev, R., Barua, S., Krechmer, J. E., Thornton, J., Ehn, M., and Kurtén, T.: Molecular mechanism for rapid autoxidation in α-pinene ozonolysis, Nat. Commun., 12, 878, https://doi.org/10.1038/s41467-021-21172-w, 2021.
Jayne, J. T., Pöschl, U., Chen, Y., Dai, D., Molina, L. T., Worsnop, D. R., Kolb, C. E., and Molina, M. J.: Pressure and temperature dependence of the gas-phase reaction of SO3 with H2O and the heterogeneous reaction of SO3 with surfaces, J. Phys. Chem. A, 101, 10000–10011, https://doi.org/10.1021/jp972549z, 1997.
Jimenez, J. L., Canagaratna, M. R., Donahue, N. M., Prevot, A. S. H., Zhang, Q., Kroll, J. H., DeCarlo, P. F., Allan, J. D., Coe, H., Ng, N. L., Aiken, A. C., Docherty, K. S., Ulbrich, I. M., Grieshop, A. P., Robinson, A. L., Duplissy, J., Smith, J. D., Wilson, K. R., Lanz, V. A., Hueglin, C., Sun, Y. L., Tian, J., Laaksonen, A., Raatikainen, T., Rautiainen, J., Vaattovaara, P., Ehn, M., Kulmala, M., Tomlinson, J. M., Collins, D. R., Cubison, M. J., E., Dunlea, J., Huffman, J. A., Onasch, T. B., Alfarra, M. R., Williams, P. I., Bower, K., Kondo, Y., Schneider, J., Drewnick, F., Borrmann, S., Weimer, S., Demerjian, K., Salcedo, D., Cottrell, L., Griffin, R., Takami, A., Miyoshi, T., Hatakeyama, S., Shimono, A., Sun, J. Y., Zhang, Y. M., Dzepina, K., Kimmel, J. R., Sueper, D., Jayne, J. T., Herndon, S. C., Trimborn, A. M., Williams, L. R., Wood, E. C., Middlebrook, A. M., Kolb, C. E., Baltensperger, U., and Worsnop, D. R.: Evolution of organic aerosols in the atmosphere, Science, 326, 1525–1529, https://doi.org/10.1126/science.1180353, 2009.
Kang, S., Wildt, J., Pullinen, I., Vereecken, L., Wu, C., Wahner, A., Zorn, S. R., and Mentel, T. F.: Formation of highly oxygenated organic molecules from α-pinene photooxidation: evidence for the importance of highly oxygenated alkoxy radicals, Atmos. Chem. Phys., 25, 15715–15740, https://doi.org/10.5194/acp-25-15715-2025, 2025.
Kangasluoma, J., Kuang, C., Wimmer, D., Rissanen, M. P., Lehtipalo, K., Ehn, M., Worsnop, D. R., Wang, J., Kulmala, M., and Petäjä, T.: Sub-3 nm particle size and composition dependent response of a nano-CPC battery, Atmos. Meas. Tech., 7, 689–700, https://doi.org/10.5194/amt-7-689-2014, 2014.
Keskinen, H., Virtanen, A., Joutsensaari, J., Tsagkogeorgas, G., Duplissy, J., Schobesberger, S., Gysel, M., Riccobono, F., Slowik, J. G., Bianchi, F., Yli-Juuti, T., Lehtipalo, K., Rondo, L., Breitenlechner, M., Kupc, A., Almeida, J., Amorim, A., Dunne, E. M., Downard, A. J., Ehrhart, S., Franchin, A., Kajos, M. K., Kirkby, J., Kürten, A., Nieminen, T., Makhmutov, V., Mathot, S., Miettinen, P., Onnela, A., Petäjä, T., Praplan, A., Santos, F. D., Schallhart, S., Sipilä, M., Stozhkov, Y., Tomé, A., Vaattovaara, P., Wimmer, D., Prevot, A., Dommen, J., Donahue, N. M., Flagan, R. C., Weingartner, E., Viisanen, Y., Riipinen, I., Hansel, A., Curtius, J., Kulmala, M., Worsnop, D. R., Baltensperger, U., Wex, H., Stratmann, F., and Laaksonen, A.: Evolution of particle composition in CLOUD nucleation experiments, Atmos. Chem. Phys., 13, 5587–5600, https://doi.org/10.5194/acp-13-5587-2013, 2013.
Kim, J., Ahlm, L., Yli-Juuti, T., Lawler, M., Keskinen, H., Tröstl, J., Schobesberger, S., Duplissy, J., Amorim, A., Bianchi, F., Donahue, N. M., Flagan, R. C., Hakala, J., Heinritzi, M., Jokinen, T., Kürten, A., Laaksonen, A., Lehtipalo, K., Miettinen, P., Petäjä, T., Rissanen, M. P., Rondo, L., Sengupta, K., Simon, M., Tomé, A., Williamson, C., Wimmer, D., Winkler, P. M., Ehrhart, S., Ye, P., Kirkby, J., Curtius, J., Baltensperger, U., Kulmala, M., Lehtinen, K. E. J., Smith, J. N., Riipinen, I., and Virtanen, A.: Hygroscopicity of nanoparticles produced from homogeneous nucleation in the CLOUD experiments, Atmos. Chem. Phys., 16, 293–304, https://doi.org/10.5194/acp-16-293-2016, 2016.
Kirkby, J., Duplissy, J., Sengupta, K., Frege, C., Gordon, H., Williamson, C., Heinritzi, M., Simon, M., Yan, C., Almeida, J., Tröstl, J., Nieminen, T., Ortega, I. K., Wagner, R., Adamov, A., Amorim, A., Bernhammer, A.-K., Bianchi, F., Breitenlechner, M., Brilke, S., Chen, X., Craven, J., Dias, A., Ehrhart, S., Flagan, R. C., Franchin, A., Fuchs, C., Guida, R., Hakala, J., Hoyle, C. R., Jokinen, T., Junninen, H., Kangasluoma, J., Kim, J., Krapf, M., Kürten, A., Laaksonen, A., Lehtipalo, K., Makhmutov, V., Mathot, S., Molteni, U., Onnela, A., Peräkylä, O., Piel, F., Petäjä, T., Praplan, A. P., Pringle, K., Rap, A., Richards, N. A. D., Riipinen, I., Rissanen, M. P., Rondo, L., Sarnela, N., Schobesberger, S., Scott, C. E., Seinfeld, J. H., Sipilä, M., Steiner, G., Stozhkov, Y., Stratmann, F., Tomé, A., Virtanen, A., Vogel, A. L., Wagner, A. C., Wagner, P. E., Weingartner, E., Wimmer, D., Winkler, P. M., Ye, P., Zhang, X., Hansel, A., Dommen, J., Donahue, N. M., Worsnop, D. R., Baltensperger, U., Kulmala, M., Carslaw, K. S., and Curtius, J.: Ion-induced nucleation of pure biogenic particles, Nature, 533, 521–526, https://doi.org/10.1038/nature17953, 2016.
Kirkby, J., Amorim, A., Baltensperger, U., Carslaw, K. S., Christoudias, T., Curtius, J., Donahue, N. M., Haddad, I. E., Flagan, R. C., Gordon, H., Hansel, A., Harder, H., Junninen, H., Kulmala, M., Kürten, A., Laaksonen, A., Lehtipalo, K., Lelieveld, J., Möhler, O., Riipinen, I., Stratmann, F., Tomé, A., Virtanen, A., Volkamer, R., Winkler, P. M., and Worsnop, D. R.: Atmospheric new particle formation from the CERN CLOUD experiment, Nat. Geosci., 16, 948–957, https://doi.org/10.1038/s41561-023-01305-0, 2023.
Kuang, C., Riipinen, I., Sihto, S.-L., Kulmala, M., McCormick, A. V., and McMurry, P. H.: An improved criterion for new particle formation in diverse atmospheric environments, Atmos. Chem. Phys., 10, 8469–8480, https://doi.org/10.5194/acp-10-8469-2010, 2010.
Kulmala, M.: How particles nucleate and grow, Science, 302, 1000–1001, https://doi.org/10.1126/science.1090848, 2003.
Kulmala, M., Laaksonen, A., and Pirjola, L.: Parameterizations for sulfuric acid/water nucleation rates, J. Geophys. Res.-Atmos., 103, 8301–8307, https://doi.org/10.1029/97JD03718, 1998.
Kulmala, M., Mordas, G., Petäjä, T., Grönholm, T., Aalto, P. P., Vehkamäki, H., Hienola, A. I., Herrmann, E., Sipilä, M., Riipinen, I., Manninen, H. E., Hämeri, K., Stratmann, F., Bilde, M., Winkler, P. M., Birmili, W., and Wagner, P. E.: The condensation particle counter battery (CPCB): A new tool to investigate the activation properties of nanoparticles, J. Aerosol. Sci., 38, 289–304, https://doi.org/10.1016/j.jaerosci.2006.11.008, 2007.
Kulmala, M., Kontkanen, J., Junninen, H., Lehtipalo, K., Manninen, H. E., Nieminen, T., Petäjä, T., Sipilä, M., Schobesberger, S., Rantala, P., Franchin, A., Jokinen, T., Järvinen, E., Äijälä, M., Kangasluoma, J., Hakala, J., Aalto, P. P., Paasonen, P., Mikkilä, J., Vanhanen, J., Aalto, J., Hakola, H., Makkonen, U., Ruuskanen, T., Mauldin, R. L., Duplissy, J., Vehkamäki, H., Bäck, J., Kortelainen, A., Riipinen, I., Kurtén, T., Johnston, M. V., Smith, J. N., Ehn, M., Mentel, T. F., Lehtinen, K. E. J., Laaksonen, A., Kerminen, V.-M., and Worsnop, D. R.: Direct observations of atmospheric aerosol nucleation, Science, 339, 943–946, https://doi.org/10.1126/science.1227385, 2013.
Kulmala, M., Petäjä, T., Ehn, M., Thornton, J., Sipilä, M., Worsnop, D. R., and Kerminen, V.-M.: Chemistry of atmospheric nucleation: On the recent advances on precursor characterization and atmospheric cluster composition in connection with atmospheric new particle formation, Annu. Rev. Phys. Chem., 65, 21–37, https://doi.org/10.1146/annurev-physchem-040412-110014, 2014.
Kurtén, T., Noppel, M., Vehkamäki, H., Salonen, M., and Kulmala, M.: Quantum chemical studies of hydrate formation of H2SO4 and HSO4– - ProQuest, Boreal Environ. Res., https://www.proquest.com/scholarly-journals/quantum-chemical-studies-hydrate-formation-h2so4/docview/2676585222/se-2 (last access: 11 February 2026), 2007.
Lee, S., Gordon, H., Yu, H., Lehtipalo, K., Haley, R., Li, Y., and Zhang, R.: New Particle Formation in the Atmosphere: From Molecular Clusters to Global Climate, J. Geophys. Res.-Atmos., 124, 7098–7146, https://doi.org/10.1029/2018JD029356, 2019.
Li, X., Li, Y., Lawler, M. J., Hao, J., Smith, J. N., and Jiang, J.: Composition of ultrafine particles in urban beijing: Measurement using a thermal desorption chemical ionization mass spectrometer, Environ. Sci. Technol., 55, 2859–2868, https://doi.org/10.1021/acs.est.0c06053, 2021.
Li, X., Li, Y., Cai, R., Yan, C., Qiao, X., Guo, Y., Deng, C., Yin, R., Chen, Y., Li, Y., Yao, L., Sarnela, N., Zhang, Y., Petäjä, T., Bianchi, F., Liu, Y., Kulmala, M., Hao, J., Smith, J. N., and Jiang, J.: Insufficient condensable organic vapors lead to slow growth of new particles in an urban environment, Environ. Sci. Technol., 56, 9936–9946, https://doi.org/10.1021/acs.est.2c01566, 2022.
Lovejoy, E. R., Hanson, D. R., and Huey, L. G.: Kinetics and products of the gas-phase reaction of SO3 with water, J. Phys. Chem., 100, 19911–19916, https://doi.org/10.1021/jp962414d, 1996.
Luo, H., Guo, Y., Shen, H., Huang, D. D., Zhang, Y., and Zhao, D.: Effect of relative humidity on the molecular composition of secondary organic aerosols from α-pinene ozonolysis, Environ. Sci.: Atmos., 4, 519–530, https://doi.org/10.1039/D3EA00149K, 2024.
Massling, A., Lange, R., Pernov, J. B., Gosewinkel, U., Sørensen, L.-L., and Skov, H.: Measurement report: High Arctic aerosol hygroscopicity at sub- and supersaturated conditions during spring and summer, Atmos. Chem. Phys., 23, 4931–4953, https://doi.org/10.5194/acp-23-4931-2023, 2023.
Massoli, P., Lambe, A. T., Ahern, A. T., Williams, L. R., Ehn, M., Mikkilä, J., Canagaratna, M. R., Brune, W. H., Onasch, T. B., Jayne, J. T., Petäjä, T., Kulmala, M., Laaksonen, A., Kolb, C. E., Davidovits, P., and Worsnop, D. R.: Relationship between aerosol oxidation level and hygroscopic properties of laboratory generated secondary organic aerosol (SOA) particles, Geophys. Res. Lett., 37, 2010GL045258, https://doi.org/10.1029/2010GL045258, 2010.
Matsubara, H., Ebisuzaki, T., and Yasuoka, K.: Microscopic insights into nucleation in a sulfuric acid–water vapor mixture based on molecular dynamics simulation, J. Chem. Phys., 130, 104705, https://doi.org/10.1063/1.3082079, 2009.
O'Dowd, C. D., Aalto, P., Hmeri, K., Kulmala, M., and Hoffmann, T.: Atmospheric particles from organic vapours, Nature, 416, 497–498, https://doi.org/10.1038/416497a, 2002.
Petters, M. D. and Kreidenweis, S. M.: A single parameter representation of hygroscopic growth and cloud condensation nucleus activity, Atmos. Chem. Phys., 7, 1961–1971, https://doi.org/10.5194/acp-7-1961-2007, 2007.
Pöhlker, M. L., Pöhlker, C., Quaas, J., Mülmenstädt, J., Pozzer, A., Andreae, M. O., Artaxo, P., Block, K., Coe, H., Ervens, B., Gallimore, P., Gaston, C. J., Gunthe, S. S., Henning, S., Herrmann, H., Krüger, O. O., McFiggans, G., Poulain, L., Raj, S. S., Reyes-Villegas, E., Royer, H. M., Walter, D., Wang, Y., and Pöschl, U.: Global organic and inorganic aerosol hygroscopicity and its effect on radiative forcing, Nat. Commun., 14, 6139, https://doi.org/10.1038/s41467-023-41695-8, 2023.
Poulain, L., Wu, Z., Petters, M. D., Wex, H., Hallbauer, E., Wehner, B., Massling, A., Kreidenweis, S. M., and Stratmann, F.: Towards closing the gap between hygroscopic growth and CCN activation for secondary organic aerosols – Part 3: Influence of the chemical composition on the hygroscopic properties and volatile fractions of aerosols, Atmos. Chem. Phys., 10, 3775–3785, https://doi.org/10.5194/acp-10-3775-2010, 2010.
Razafindrambinina, P. N., Malek, K. A., Dawson, J. N., DiMonte, K., Raymond, T. M., Dutcher, D. D., Freedman, M. A., and Asa-Awuku, A.: Hygroscopicity of internally mixed ammonium sulfate and secondary organic aerosol particles formed at low and high relative humidity, Environ. Sci.: Atmos., 2, 202–214, https://doi.org/10.1039/D1EA00069A, 2022.
Ren, J., Chen, L., Fan, T., Liu, J., Jiang, S., and Zhang, F.: The NPF effect on CCN number concentrations: A review and re-evaluation of observations from 35 sites worldwide, Geophys. Res. Lett., 48, e2021GL095190, https://doi.org/10.1029/2021GL095190, 2021.
Riccobono, F., Rondo, L., Sipilä, M., Barmet, P., Curtius, J., Dommen, J., Ehn, M., Ehrhart, S., Kulmala, M., Kürten, A., Mikkilä, J., Paasonen, P., Petäjä, T., Weingartner, E., and Baltensperger, U.: Contribution of sulfuric acid and oxidized organic compounds to particle formation and growth, Atmos. Chem. Phys., 12, 9427–9439, https://doi.org/10.5194/acp-12-9427-2012, 2012.
Riccobono, F., Schobesberger, S., Scott, C. E., Dommen, J., Ortega, I. K., Rondo, L., Almeida, J., Amorim, A., Bianchi, F., Breitenlechner, M., David, A., Downard, A., Dunne, E. M., Duplissy, J., Ehrhart, S., Flagan, R. C., Franchin, A., Hansel, A., Junninen, H., Kajos, M., Keskinen, H., Kupc, A., Kürten, A., Kvashin, A. N., Laaksonen, A., Lehtipalo, K., Makhmutov, V., Mathot, S., Nieminen, T., Onnela, A., Petäjä, T., Praplan, A. P., Santos, F. D., Schallhart, S., Seinfeld, J. H., Sipilä, M., Spracklen, D. V., Stozhkov, Y., Stratmann, F., Tomé, A., Tsagkogeorgas, G., Vaattovaara, P., Viisanen, Y., Vrtala, A., Wagner, P. E., Weingartner, E., Wex, H., Wimmer, D., Carslaw, K. S., Curtius, J., Donahue, N. M., Kirkby, J., Kulmala, M., Worsnop, D. R., and Baltensperger, U.: Oxidation products of biogenic emissions contribute to nucleation of atmospheric particles, Science, 344, 717–721, https://doi.org/10.1126/science.1243527, 2014.
Riipinen, I., Yli-Juuti, T., Pierce, J. R., Petäjä, T., Worsnop, D. R., Kulmala, M., and Donahue, N. M.: The contribution of organics to atmospheric nanoparticle growth, Nat. Geosci., 5, 453–458, https://doi.org/10.1038/ngeo1499, 2012.
Shantz, N. C., Leaitch, W. R., Phinney, L., Mozurkewich, M., and Toom-Sauntry, D.: The effect of organic compounds on the growth rate of cloud droplets in marine and forest settings, Atmos. Chem. Phys., 8, 5869–5887, https://doi.org/10.5194/acp-8-5869-2008, 2008.
Sipilä, M., Berndt, T., Petäjä, T., Brus, D., Vanhanen, J., Stratmann, F., Patokoski, J., Mauldin, R. L., Hyvärinen, A.-P., Lihavainen, H., and Kulmala, M.: The role of sulfuric acid in atmospheric nucleation, Science, 327, 1243–1246, https://doi.org/10.1126/science.1180315, 2010.
Smith, J. N., Draper, D. C., Chee, S., Dam, M., Glicker, H., Myers, D., Thomas, A. E., Lawler, M. J., and Myllys, N.: Atmospheric clusters to nanoparticles: Recent progress and challenges in closing the gap in chemical composition, J. Aerosol. Sci., 153, 105733, https://doi.org/10.1016/j.jaerosci.2020.105733, 2021.
Steiner, G.: High resolution mobility spectrometry of molecular ions and their effect on the charging probabilities of airborne particles under bipolar diffusion charging conditions, PhD thesis, University of Vienna, Vienna, Austria, https://doi.org/10.25365/thesis.14750, 2011.
Stolzenburg, D., Fischer, L., Vogel, A. L., Heinritzi, M., Schervish, M., Simon, M., Wagner, A. C., Dada, L., Ahonen, L. R., Amorim, A., Baccarini, A., Bauer, P. S., Baumgartner, B., Bergen, A., Bianchi, F., Breitenlechner, M., Brilke, S., Buenrostro Mazon, S., Chen, D., Dias, A., Draper, D. C., Duplissy, J., El Haddad, I., Finkenzeller, H., Frege, C., Fuchs, C., Garmash, O., Gordon, H., He, X., Helm, J., Hofbauer, V., Hoyle, C. R., Kim, C., Kirkby, J., Kontkanen, J., Kürten, A., Lampilahti, J., Lawler, M., Lehtipalo, K., Leiminger, M., Mai, H., Mathot, S., Mentler, B., Molteni, U., Nie, W., Nieminen, T., Nowak, J. B., Ojdanic, A., Onnela, A., Passananti, M., Petäjä, T., Quéléver, L. L. J., Rissanen, M. P., Sarnela, N., Schallhart, S., Tauber, C., Tomé, A., Wagner, R., Wang, M., Weitz, L., Wimmer, D., Xiao, M., Yan, C., Ye, P., Zha, Q., Baltensperger, U., Curtius, J., Dommen, J., Flagan, R. C., Kulmala, M., Smith, J. N., Worsnop, D. R., Hansel, A., Donahue, N. M., and Winkler, P. M.: Rapid growth of organic aerosol nanoparticles over a wide tropospheric temperature range, Proc. Natl. Acad. Sci. USA, 115, 9122–9127, https://doi.org/10.1073/pnas.1807604115, 2018.
Stolzenburg, D., Simon, M., Ranjithkumar, A., Kürten, A., Lehtipalo, K., Gordon, H., Ehrhart, S., Finkenzeller, H., Pichelstorfer, L., Nieminen, T., He, X.-C., Brilke, S., Xiao, M., Amorim, A., Baalbaki, R., Baccarini, A., Beck, L., Bräkling, S., Caudillo Murillo, L., Chen, D., Chu, B., Dada, L., Dias, A., Dommen, J., Duplissy, J., El Haddad, I., Fischer, L., Gonzalez Carracedo, L., Heinritzi, M., Kim, C., Koenig, T. K., Kong, W., Lamkaddam, H., Lee, C. P., Leiminger, M., Li, Z., Makhmutov, V., Manninen, H. E., Marie, G., Marten, R., Müller, T., Nie, W., Partoll, E., Petäjä, T., Pfeifer, J., Philippov, M., Rissanen, M. P., Rörup, B., Schobesberger, S., Schuchmann, S., Shen, J., Sipilä, M., Steiner, G., Stozhkov, Y., Tauber, C., Tham, Y. J., Tomé, A., Vazquez-Pufleau, M., Wagner, A. C., Wang, M., Wang, Y., Weber, S. K., Wimmer, D., Wlasits, P. J., Wu, Y., Ye, Q., Zauner-Wieczorek, M., Baltensperger, U., Carslaw, K. S., Curtius, J., Donahue, N. M., Flagan, R. C., Hansel, A., Kulmala, M., Lelieveld, J., Volkamer, R., Kirkby, J., and Winkler, P. M.: Enhanced growth rate of atmospheric particles from sulfuric acid, Atmos. Chem. Phys., 20, 7359–7372, https://doi.org/10.5194/acp-20-7359-2020, 2020.
Stolzenburg, D., Cai, R., Blichner, S. M., Kontkanen, J., Zhou, P., Makkonen, R., Kerminen, V.-M., Kulmala, M., Riipinen, I., and Kangasluoma, J.: Atmospheric nanoparticle growth, Rev. Mod. Phys., 95, 045002, https://doi.org/10.1103/RevModPhys.95.045002, 2023.
Sullivan, R. C., Petters, M. D., DeMott, P. J., Kreidenweis, S. M., Wex, H., Niedermeier, D., Hartmann, S., Clauss, T., Stratmann, F., Reitz, P., Schneider, J., and Sierau, B.: Irreversible loss of ice nucleation active sites in mineral dust particles caused by sulphuric acid condensation, Atmos. Chem. Phys., 10, 11471–11487, https://doi.org/10.5194/acp-10-11471-2010, 2010.
Sun, J., Hermann, M., Weinhold, K., Merkel, M., Birmili, W., Yang, Y., Tuch, T., Flentje, H., Briel, B., Ries, L., Couret, C., Elsasser, M., Sohmer, R., Wirtz, K., Meinhardt, F., Schütze, M., Bath, O., Hellack, B., Kerminen, V.-M., Kulmala, M., Ma, N., and Wiedensohler, A.: Measurement report: Contribution of atmospheric new particle formation to ultrafine particle concentration, cloud condensation nuclei, and radiative forcing – results from 5-year observations in central Europe, Atmos. Chem. Phys., 24, 10667–10687, https://doi.org/10.5194/acp-24-10667-2024, 2024.
Surdu, M., Lamkaddam, H., Wang, D. S., Bell, D. M., Xiao, M., Lee, C. P., Li, D., Caudillo, L., Marie, G., Scholz, W., Wang, M., Lopez, B., Piedehierro, A. A., Ataei, F., Baalbaki, R., Bertozzi, B., Bogert, P., Brasseur, Z., Dada, L., Duplissy, J., Finkenzeller, H., He, X.-C., Höhler, K., Korhonen, K., Krechmer, J. E., Lehtipalo, K., Mahfouz, N. G. A., Manninen, H. E., Marten, R., Massabò, D., Mauldin, R., Petäjä, T., Pfeifer, J., Philippov, M., Rörup, B., Simon, M., Shen, J., Umo, N. S., Vogel, F., Weber, S. K., Zauner-Wieczorek, M., Volkamer, R., Saathoff, H., Möhler, O., Kirkby, J., Worsnop, D. R., Kulmala, M., Stratmann, F., Hansel, A., Curtius, J., Welti, A., Riva, M., Donahue, N. M., Baltensperger, U., and El Haddad, I.: Molecular understanding of the enhancement in organic aerosol mass at high relative humidity, Environ. Sci. Technol., 57, 2297–2309, https://doi.org/10.1021/acs.est.2c04587, 2023.
Temelso, B., Morrell, T. E., Shields, R. M., Allodi, M. A., Wood, E. K., Kirschner, K. N., Castonguay, T. C., Archer, K. A., and Shields, G. C.: Quantum mechanical study of sulfuric acid hydration: Atmospheric implications, J. Phys. Chem. A, 116, 2209–2224, https://doi.org/10.1021/jp2119026, 2012.
Tröstl, J., Chuang, W. K., Gordon, H., Heinritzi, M., Yan, C., Molteni, U., Ahlm, L., Frege, C., Bianchi, F., Wagner, R., Simon, M., Lehtipalo, K., Williamson, C., Craven, J. S., Duplissy, J., Adamov, A., Almeida, J., Bernhammer, A.-K., Breitenlechner, M., Brilke, S., Dias, A., Ehrhart, S., Flagan, R. C., Franchin, A., Fuchs, C., Guida, R., Gysel, M., Hansel, A., Hoyle, C. R., Jokinen, T., Junninen, H., Kangasluoma, J., Keskinen, H., Kim, J., Krapf, M., Kürten, A., Laaksonen, A., Lawler, M., Leiminger, M., Mathot, S., Möhler, O., Nieminen, T., Onnela, A., Petäjä, T., Piel, F. M., Miettinen, P., Rissanen, M. P., Rondo, L., Sarnela, N., Schobesberger, S., Sengupta, K., Sipilä, M., Smith, J. N., Steiner, G., Tomè, A., Virtanen, A., Wagner, A. C., Weingartner, E., Wimmer, D., Winkler, P. M., Ye, P., Carslaw, K. S., Curtius, J., Dommen, J., Kirkby, J., Kulmala, M., Riipinen, I., Worsnop, D. R., Donahue, N. M., and Baltensperger, U.: The role of low-volatility organic compounds in initial particle growth in the atmosphere, Nature, 533, 527–531, https://doi.org/10.1038/nature18271, 2016.
Vogel, A. L., Schneider, J., Müller-Tautges, C., Phillips, G. J., Pöhlker, M. L., Rose, D., Zuth, C., Makkonen, U., Hakola, H., Crowley, J. N., Andreae, M. O., Pöschl, U., and Hoffmann, T.: Aerosol chemistry resolved by mass spectrometry: Linking field measurements of cloud condensation nuclei activity to organic aerosol composition, Environ. Sci. Technol., 50, 10823–10832, https://doi.org/10.1021/acs.est.6b01675, 2016.
Wang, J., Shilling, J. E., Liu, J., Zelenyuk, A., Bell, D. M., Petters, M. D., Thalman, R., Mei, F., Zaveri, R. A., and Zheng, G.: Cloud droplet activation of secondary organic aerosol is mainly controlled by molecular weight, not water solubility, Atmos. Chem. Phys., 19, 941–954, https://doi.org/10.5194/acp-19-941-2019, 2019.
Wang, Z., Su, H., Wang, X., Ma, N., Wiedensohler, A., Pöschl, U., and Cheng, Y.: Scanning supersaturation condensation particle counter applied as a nano-CCN counter for size-resolved analysis of the hygroscopicity and chemical composition of nanoparticles, Atmos. Meas. Tech., 8, 2161–2172, https://doi.org/10.5194/amt-8-2161-2015, 2015.
Wang, Z. B., Hu, M., Yue, D. L., Zheng, J., Zhang, R. Y., Wiedensohler, A., Wu, Z. J., Nieminen, T., and Boy, M.: Evaluation on the role of sulfuric acid in the mechanisms of new particle formation for Beijing case, Atmos. Chem. Phys., 11, 12663–12671, https://doi.org/10.5194/acp-11-12663-2011, 2011.
Wiedensohler, A. and Fissan, H. J.: Aerosol charging in high purity gases, J. Aerosol Sci., 19, 867–870, https://doi.org/10.1016/0021-8502(88)90054-7, 1988.
Wiedensohler, A., Lütkemeier, E., Feldpausch, M., and Helsper, C.: Investigation of the bipolar charge distribution at various gas conditions, Journal of Aerosol Science, 17, 413–416, https://doi.org/10.1016/0021-8502(86)90118-7, 1986.
Xiao, S., Wang, M. Y., Yao, L., Kulmala, M., Zhou, B., Yang, X., Chen, J. M., Wang, D. F., Fu, Q. Y., Worsnop, D. R., and Wang, L.: Strong atmospheric new particle formation in winter in urban Shanghai, China, Atmos. Chem. Phys., 15, 1769–1781, https://doi.org/10.5194/acp-15-1769-2015, 2015.
Yang, H., Raucci, U., Iyer, S., Hasan, G., Golin Almeida, T., Barua, S., Savolainen, A., Kangasluoma, J., Rissanen, M., Vehkamäki, H., and Kurtén, T.: Molecular dynamics-guided reaction discovery reveals endoperoxide-to-alkoxy radical isomerization as key branching point in α-pinene ozonolysis, Nat. Commun., 16, 661, https://doi.org/10.1038/s41467-025-55985-w, 2025.
Yao, L., Garmash, O., Bianchi, F., Zheng, J., Yan, C., Kontkanen, J., Junninen, H., Mazon, S. B., Ehn, M., Paasonen, P., Sipilä, M., Wang, M., Wang, X., Xiao, S., Chen, H., Lu, Y., Zhang, B., Wang, D., Fu, Q., Geng, F., Li, L., Wang, H., Qiao, L., Yang, X., Chen, J., Kerminen, V.-M., Petäjä, T., Worsnop, D. R., Kulmala, M., and Wang, L.: Atmospheric new particle formation from sulfuric acid and amines in a Chinese megacity, Science, 361, 278–281, https://doi.org/10.1126/science.aao4839, 2018.
Yishake, J., Zang, H., Tan, R., Yao, L., Guo, S., Zhao, Y., and Li, C.: Size- and Composition-Dependent Hygroscopic Growth of Sub-20 nm Atmospherically Relevant Particles: Implications for New Particle Survival, Environ. Sci. Technol., acs.est.5c00068, https://doi.org/10.1021/acs.est.5c00068, 2025.
Yu, H., Dai, L., Zhao, Y., Kanawade, V. P., Tripathi, S. N., Ge, X., Chen, M., and Lee, S.: Laboratory observations of temperature and humidity dependencies of nucleation and growth rates of sub-3 nm particles, J. Geophys. Res.-Atmos., 122, 1919–1929, https://doi.org/10.1002/2016JD025619, 2017.
Yuan, C., Ma, Y., Diao, Y., Yao, L., Zhou, Y., Wang, X., and Zheng, J.: CCN activity of secondary aerosols from terpene ozonolysis under atmospheric relevant conditions, J. Geophys. Res.-Atmos., 122, 4654–4669, https://doi.org/10.1002/2016JD026039, 2017.
Zhang, K., Xu, Z., Gao, J., Xu, Z., and Wang, Z.: Review of online measurement techniques for chemical composition of atmospheric clusters and sub-20 nm particles, Front. Environ. Sci., 10, 937006, https://doi.org/10.3389/fenvs.2022.937006, 2022.
Zhang, K., Xu, Z., Pei, X., Zhang, F., Su, H., Cheng, Y., and Wang, Z.: Characteristics of scanning flow condensation particle counter (SFCPC): A rapid approach for retrieving hygroscopicity and chemical composition of sub-10 nm aerosol particles, Aerosol Sci. Technol., 57, 1031–1043, https://doi.org/10.1080/02786826.2023.2245859, 2023.
Zhang, K., Xiong, C., Cheng, Y., Ma, N., Mikhailov, E., Pöschl, U., Su, H., and Wang, Z.: Assessment of Hygroscopicity Uncertainties Associated With Size and Thermodynamic Model: Implications for Inferring Chemical Composition of Sub-10 nm Particles, J. Geophys. Res.-Atmos., 130, e2025JD043835, https://doi.org/10.1029/2025JD043835, 2025.
Zhang, R., Suh, I., Zhao, J., Zhang, D., Fortner, E. C., Tie, X., Molina, L. T., and Molina, M. J.: Atmospheric new particle formation enhanced by organic acids, Science, 304, 1487–1490, https://doi.org/10.1126/science.1095139, 2004.
Zhang, R., Wang, L., Khalizov, A. F., Zhao, J., Zheng, J., McGraw, R. L., and Molina, L. T.: Formation of nanoparticles of blue haze enhanced by anthropogenic pollution, Proc. Natl. Acad. Sci. USA, 106, 17650–17654, https://doi.org/10.1073/pnas.0910125106, 2009.
Zhang, R., Khalizov, A., Wang, L., Hu, M., and Xu, W.: Nucleation and growth of nanoparticles in the atmosphere, Chem. Rev., 112, 1957–2011, https://doi.org/10.1021/cr2001756, 2012.
Zhao, B., Donahue, N. M., Zhang, K., Mao, L., Shrivastava, M., Ma, P.-L., Shen, J., Wang, S., Sun, J., Gordon, H., Tang, S., Fast, J., Wang, M., Gao, Y., Yan, C., Singh, B., Li, Z., Huang, L., Lou, S., Lin, G., Wang, H., Jiang, J., Ding, A., Nie, W., Qi, X., Chi, X., and Wang, L.: Global variability in atmospheric new particle formation mechanisms, Nature, 631, 98–105, https://doi.org/10.1038/s41586-024-07547-1, 2024.
Zhao, D. F., Buchholz, A., Kortner, B., Schlag, P., Rubach, F., Fuchs, H., Kiendler-Scharr, A., Tillmann, R., Wahner, A., Watne, Å. K., Hallquist, M., Flores, J. M., Rudich, Y., Kristensen, K., Hansen, A. M. K., Glasius, M., Kourtchev, I., Kalberer, M., and Mentel, Th. F.: Cloud condensation nuclei activity, droplet growth kinetics, and hygroscopicity of biogenic and anthropogenic secondary organic aerosol (SOA), Atmos. Chem. Phys., 16, 1105–1121, https://doi.org/10.5194/acp-16-1105-2016, 2016.
Zhou, R., Afsana, S., Wei, C., and Mochida, M.: Additive water uptake of the mixtures of urban atmospheric HULIS and ammonium sulfate, J. Geophys. Res.-Atmos., 129, e2023JD040553, https://doi.org/10.1029/2023JD040553, 2024.
Zollner, J. H., Glasoe, W. A., Panta, B., Carlson, K. K., McMurry, P. H., and Hanson, D. R.: Sulfuric acid nucleation: power dependencies, variation with relative humidity, and effect of bases, Atmos. Chem. Phys., 12, 4399–4411, https://doi.org/10.5194/acp-12-4399-2012, 2012.