the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Water activity and surface tension of aqueous ammonium sulfate and D-glucose aerosol nanoparticles
Eugene F. Mikhailov
Sergey S. Vlasenko
Water activity (aw) and interfacial energy or surface tension (σ) are key thermodynamic parameters to describe the hygroscopic growth of atmospheric aerosol particles and their ability to serve as cloud condensation nuclei (CCN), thus influencing the hydrological cycle and climate. Due to size effects and complex mixing states, however, these parameters are not well constrained for nanoparticles composed of organic and inorganic compounds in aqueous solution.
In this study, we determined aw and σ by differential Köhler analysis (DKA) of hygroscopic growth measurement data for aerosol particles smaller than 100 nm composed of aqueous ammonium sulfate (AS), D-glucose (Gl), and their mixtures. High-precision measurements of hygroscopic growth were performed at relative humidities (denoted RH) ranging from 2.0 % to 99.6 % with a high-humidity tandem differential mobility analyzer (HHTDMA) in three complementary modes of operation: hydration, dehydration, and restructuring. The restructuring mode (hydration followed by dehydration) enabled the transformation of initially irregular particles into compact globules and the determination of mass equivalent diameters. The HHTDMA-derived growth factors complemented by DKA allows for determination of water activity and surface tension from dilute to highly supersaturated aqueous solutions that are not accessible with other methods. Thus, for mixed AS / Gl nanoparticles with mass ratios of 4:1 and 1:1, the upper limit of solute mass fraction (Xs) was 0.92 and 0.98, respectively.
For pure AS and Gl, the DKA-derived aw is in good agreement with electrodynamic balance and bulk measurement data. For AS particles, our aw data also agree well with the Extended Aerosol Inorganics Model (E-AIM III) over the entire concentration range. In contrast, the UNIFAC model as a part of AIOMFAC (Zuend et al., 2011) was found to overestimate aw in aqueous Gl particles, which can be attributed to unaccounted intermolecular interactions.
For mixed AS and Gl nanoparticles, we observed a non-monotonic concentration dependence of the surface tension that does not follow the predictions by modeling approaches constructed for mixed inorganic/organic systems. Thus, AS / Gl particles with a 1:1 mass ratio exhibited a strong decrease of σ with increasing solute mass fraction, a minimum value of 56.5 mN m−1 at Xs≈0.5, and a reverse trend of increasing σ at higher concentrations. We suggest that D-glucose molecules surrounded by ammonium sulfate ions tend to associate, forming non-polar aggregates, which lowers the surface tension at the air–droplet interface.
We analyzed the uncertainty in the DKA-derived water activity and surface tension, related to the instrumental errors as well as to the morphology of the nanoparticles and their phase state. Our studies have shown that under optimal modes of operation of HHTDMA for moderate aqueous concentrations, the uncertainty in aw and σ does not exceed 0.2 %–0.4 % and 3 %–4 %, respectively, but it increases by an order of magnitude in the case of highly concentrated nanodroplet solutions.
- Article
(4212 KB) - Full-text XML
-
Supplement
(711 KB) - BibTeX
- EndNote
Water uptake of aerosol particles is among the central issues of current research into atmospheric and climate processes (Pöschl, 2005; Andreae and Rosenfeld, 2008). Water activity and surface tension (interfacial energy) are key thermodynamic parameters of classical Köhler theory, which describes the hygroscopic growth of particles and their ability to serve as cloud condensation nuclei (CCN) (Swietlicki et al., 2008; Prisle et al., 2011; Forestieri et al., 2018; Ruehl et al., 2016; Ovadnevaite et al., 2017; Davies et al., 2019). Bulk methods, such as pendant drop tensiometry (Anastasiadis et al., 1987; Topping et al., 2007; Shapiro et al., 2009), electrodynamic balance (EDB), and optical tweezers operating with super-micrometric particles are mainly used to determine aw and σ (Tang et al., 2019; Bzdek et al., 2020). The applicability of these methods to nanoparticles is limited because the water uptake, gas–particle and bulk–surface partitioning, and related phase transitions (deliquescence, efflorescence) depend on particle size (Biskos et al., 2006a, b; Prisle et al., 2010; Djikaev et al., 2001; Cheng et al., 2015). To bridge the gap between experimental and modeling results for bulk materials and nanoparticles, a new method called differential Köhler analysis (DKA) was developed recently (Cheng et al., 2015). This method allows for determination of the water activity and surface tension of supersaturated aqueous solutions based on hygroscopic growth measurement of nano-sized droplets. Recently, Lei et al. (2023) used the DKA method to estimate water activity of the levoglucosan and D-glucose aerosol nanoparticles using nano-HTDMA (hygroscopicity tandem differential mobility analyzer) in the size range of 6–100 nm with RH below 90 %. These results show a good agreement of the DKA-derived water activity with the earlier experimental data and E-AIM. However, no DKA-based surface tension data were reported in that study.
Atmospheric aerosol particles are mainly a mixture of organic and inorganic components. The interaction of organic–inorganic species in aqueous solutions affects both bulk (Raoul effect) and surface (Kelvin effect) properties of nanoparticles. In order to estimate these effects independently, model approximations for aw or σ are introduced (Li and Lu, 2001; Tuckermann, 2007; Prisle et al., 2010; Petters and Petters, 2016; Ruehl et al., 2016; Ovadnevaite et al., 2017; Forestieri et al., 2018; Davies et al., 2019; Schmedding and Zuend, 2023; and references therein). However, it is difficult to verify the validity of these approximations with respect to the nanodroplets accounting for the bulk–surface partitioning and liquid–liquid separation effects. The advantage of the DKA method is precisely that it allows for simultaneous determination of the bulk and surface characteristics of nanodroplets in the wide concentration range, including a highly supersaturated solution. This provides a new opportunity for validation of the theoretical approaches used for both aw and σ in the pure and mixed organic–inorganic aerosols. In addition to pure substances, the DKA method is applied here to derive the water activity and surface tension of two-component particles comprising ammonium sulfate and D-glucose with mass ratios of 4:1 and 1:1. Both components are surface inactive and highly water soluble.
The DKA data were obtained from the measurements of hygroscopic growth factors with a high-humidity tandem differential mobility analyzer (HHTDMA) for aerosol particles with diameters in the range of 17–100 nm at relative humidity (RH) of 2.0 %–99.6 %. The high precision of growth factor measurements in the wide particle size and RH range allows for determination of the water activity and surface tension from dilute to highly supersaturated aqueous solutions under conditions that are not accessible to other methods.
We analyze and discuss our nanoparticle measurement results with respect to those obtained with bulk experimental methods and thermodynamic models. In addition, our HHTDMA measurements provide information about particle restructuring in response to water vapor adsorption at low and intermediate RH levels (Mikhailov et al., 2009, 2021), which enable the determination of mass equivalent diameters for the investigated nanoparticles as well as uncertainty analyses and refinements of the DKA-derived thermodynamic parameters at subsaturation and supersaturation conditions.
2.1 Aerosol generation
The investigated aerosols were produced by nebulizing an aqueous solution of pure ammonium sulfate (99.9 % pure, ChemCruz) and D-glucose (99.55 % pure, Fisher) at ∼0.01 % weight concentration or their mixture with and 1:1 weight ratios.
2.2 HHTDMA setup and modes of operation
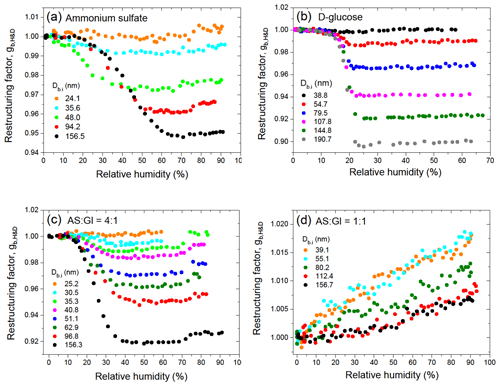
The hygroscopic properties of size-selected aerosol particles were measured in the 2.0 %–99.6 % RH range with a high-humidity tandem differential mobility analyzer (HHTDMA) (see Mikhailov and Vlasenko, 2020; Mikhailov et al., 2021). Throughout the whole relative humidity range, the absolute uncertainty of RH is less than 0.5 %, and the relative growth factor uncertainty due to RH and instrumental errors does not exceed 1 %. A detailed calculation of growth factor uncertainty is described in Mikhailov and Vlasenko (2020) (Sect. 2.5). Three operation modes are available using this HHTDMA instrument: hydration and dehydration (H&D) (also called restructuring mode), hydration, and dehydration. The H&D mode was used to determine the optimal RH range in which initial irregular particles transform into compact globules.
Inorganic and organic aerosol particles as well as their mixtures restructure upon humidification below their deliquescence (Mikhailov et al., 2020). Irregular envelope shape and porosity cause a discrepancy between the mobility-equivalent and mass-equivalent particle diameters that limits precision of the mobility-diameter-based hygroscopicity tandem differential mobility analyzer (HTDMA) (Gysel et al., 2004; Mikhailov et al., 2020). To account for restructuring, we used the minimum mobility particle diameter, Db,H&D,min, obtained in the H&D HHTDMA mode as an approximation of mass-equivalent diameter of the dry solute particle, Ds (i.e., Ds=Db,H&D,min). The size-dependent restructuring factor was calculated as follows: , where Db,i and Db,RH are the initial mobility particle diameter and that measured at RH of aerosol preconditioning, respectively (Mikhailov et al., 2021, Supplement S3). The description of the particle restructuring mechanism is beyond the scope of this work. It will be considered in detail in the next paper.
In this study, the H&D mode was in situ coupled with a conventional hydration or dehydration mode. In the combined modes, the initial monodisperse dry particles were first led through the preconditioning section where they underwent microstructural transformation, by means of humidification in the 2 %–90 % RH range (Nafion humidifier) and drying (Nafion dryer followed by a silica gel diffusion dryer). At the same time, the aerosol and sheath flow in the second differential mobility analyzer was below 3 % RH (Mikhailov et al., 2020, 2021, Supplement S3). The preconditioning RH at which the particles reached their minimum diameter, Db,H&D;min, was then kept constant throughout the standard hydration or dehydration experiment. The mobility-equivalent particle growth factor, gb, was calculated as the ratio of the mobility-equivalent diameter, Db, measured after conditioning (hydration, dehydration) to the minimum mobility diameter, Db;H&D;min, observed in the H&D mode:
3.1 Differential Köhler analysis (DKA)
The basis of the DKA method (Cheng et al., 2015) is the Köhler equation which describes the equilibrium relative humidity, sw, over a spherical droplet as a function of aw, σ, and the mass equivalent diameter Db:
where vw is the partial molar volume of water, R is the universal gas constant, and T is the temperature. Db in Eq. (2) is often replaced by the product of Ds and gb; that is, . The logarithmic form of Eq. (2) is
where . From Eq. (3) it follows that aw and σ can be obtained as the fitting parameters for the sw(Ds) dependence with the same gb. Obviously, the larger the number of experimental values of Ds that are used for the fitting, the more accurately aw and σ can be estimated. This study utilized four sizes for ammonium sulfate and eight sizes for D-glucose and its mixture with ammonium sulfate in the Ds range 17–100 nm, where the Kelvin curvature effect has a significant impact on particle hygroscopicity. We used the Levenberg–Marquardt algorithm (implemented in Origin 9.0) to find aw and σ in the iterative procedure with standard fitting error as a measure of their uncertainty. It should be noted that fitting of the experimental sw(Ds) dependences over the entire set of dry diameters significantly improves the reliability of the DKA-derived aw and σ as compared to the original approach, where these quantities were calculated in pairs for available dry diameters (Cheng et al., 2015; Lei et al., 2023).
According to Eq. (3), DKA assumes that water activity and surface tension depend on gb only (i.e., concentration). However, in the case of bulk–surface partitioning, both water activity and surface tension may also depend on Ds. To minimize this effect, we used surface inactive compounds. Previous studies on the example of NaCl and (NH4)2SO4 nanoparticles have shown that the size effect is negligible for such compounds (Bahadur and Russell, 2008; Cheng et al., 2015, Supplement).
3.2 HHTDMA-derived activity coefficients
In a binary system at a constant temperature and pressure, the water activity, aw, and the solute activity, as, are related by the Gibbs–Duhem equation (Prausnitz et al., 1999):
where xw and xs are the mole fractions of water and solute, respectively. According to Robinson and Stokes (1970), the water activity and molal osmotic coefficient of solute, Φs, for the real solution are related by
where νs and ms are the stoichiometric dissociation number and molality of the solute (mol kg−1 of water), and Mw is the molecular mass of water (g mol−1). For electrolyte solution in the molality scale,
ν+ and ν− are the numbers of positive and negative ions produced upon dissociation of the solute (); and m±, γ±, and a± are the mean molality, activity coefficient, and activity of the solute. For ammonium sulfate with νs=3 and non-dissociating D-glucose with νs=1 from Eq. (6) it follows that
Combining Eqs. (4)–(6) gives (Prausnitz et al., 1999)
Integration from ms=0 to the solution of interest yields
The integral in Eq. (9) was evaluated numerically by plotting values of against ms.
3.3 Surface tension models
The DKA-derived σ of pure AS and Gl aqueous solutions and their mixtures were compared with the model of Li and Lu (2001) and Dutcher et al. (2010). These models were moderately successful at representing the surface tensions of aqueous solutions of some soluble salts and their mixtures with organic compounds (Topping et al., 2007). The model by Dutcher et al. (2010) expresses the surface tension (σ) in terms of the surface tension of pure water (σw) and that of a hypothetical molten salt (σs):
where xw and xs are the mole fractions of water (w) and salt (s). It is assumed that the surface tension of a solution at different temperatures is described by , with , where aws and bws are fitted parameters. At very high salt concentrations, a similar relationship is used: with . The two above expressions can be applied to express the surface tension over the entire concentration range:
Equation (11) was used to calculate σ of ammonium sulfate particles at T=298 K with parameters σw=72.0 mN m−1, σs=185.0 mN m−1, , bws=0.366 , and as suggested by Dutcher et al. (2010). The Li and Lu (2001) surface tension model includes a Langmuir-type adsorption isotherm and a term dependent on the activity of dissolved solid. For an aqueous solution with one solute, this model yields
where is the saturated surface excess of solute (s), and Ks is the adsorption equilibrium constant of solute (s). For multi-component systems, Li and Lu (2001) proposed two approaches. The first approach, referred to as LiLu (1), implies that there is no interaction or competing adsorption between species at the interface:
The second approach, that we refer to as LiLu (2), considers the interacting and competing adsorption between species in the interface for mixed aqueous systems at higher concentrations. This yields another expression for the surface tension of mixed solutions:
We also compared the DKA–derived σ with simpler approximations, including those describing a supersaturated solution. Thus, for a ammonium sulfate solution, we used the parameterization by Pruppacher and Klett (1997):
where Xs is the mass fraction of solute. This parameterization is expected to be valid up to Xs≈0.8. For the concentration dependence of surface tension of an aqueous D-glucose solution, the linear function given by Aumann et al. (2010) was chosen:
where C is the molarity concentration (mol L−1) and (mN m−1/mol L−1) with upper limit of C being 3.5 mol L−1 (mGl=6.0 mol kg−1).
4.1 Particle restructuring due to humidification
Figure 1 illustrates the restructuring of aerosol particles in the H&D mode. Both pure and mixed particles restructure with increasing RH, wherein this effect is strongest for the largest diameters. Interestingly, mixed AS / Gl particles with a mass ratio of 4:1 undergo strong restructuring with gb,H&D less than 1 (Fig. 1c), whereas AS / Gl particles with a mass ratio of 1:1 show no structural change (Fig. 1d). For these particles, the restructuring factor gb,H&D is larger than 1 and increases with RH, indicating that initial particles are compact and spherical (i.e., ) (Mikhailov et al., 2020). The size-dependent increase in gb,H&D (Fig. 1c) is due to a residual water film on the particle surface after dehydration in the H&D mode. The higher values of observed for particles with smaller diameters support this assumption. Since the H&D mode is in situ coupled with a conventional hydration or dehydration mode, the observed Db,H&D,min values at specified RH were directly used to calculate the growth factor using Eq. (1). This has made it possible to eliminate the uncertainty in the growth factor caused by irregular particle morphology, which is as high as 5 % or more for particles with Db,i>70 nm.
4.2 Size-dependent growth factors
Figure 2 shows the size-dependent growth factors of pure and mixed particles observed upon dehydration in the HHTDMA mode, calculated using Eq. (1). The dehydration branch was used because it allows us to determine aw and σ over a wide concentration range, including highly concentrated droplet solutions. Note that, in contrast to pure ammonium sulfate and its 4:1 mixture with D-glucose, in the case of pure D-glucose (Fig. 2b) and mixed AS / Gl particles with a mass ratio of 1:1 (Fig. 2d), the growth factors obtained in the hydration and dehydration modes are identical over the whole range of relative humidity (Fig. S1 in the Supplement). As expected, due to the Kelvin effect, for the same gb the equilibrium values of sw (or RH) for smaller particles shift towards higher values (Eq. 2). This is clearly visible for all particles measured at RH above 70 % (inserts in Fig. 2). For ammonium sulfate particles, this pattern is valid also for highly concentrated solutions up to efflorescence RH (ERH ∼30 %) (Fig. 3a). However, in the cases of D-glucose (Fig. 3b) and AS / Gl (1:1) mixed particles (Fig. 3d), this pattern is not obvious. Moreover, in the case of AS / Gl (4:1) particles, the values of sw for small particles are lower than for large particles (Fig. 3c), in contrast to the behavior of pure ammonium sulfate (Fig. 3a). The measurement data in Fig. 3b and d also demonstrate that for D-glucose and AS / Gl (1:1) particles in the vicinity of RH≈48 % the gb(RH) dependence has an inflection point (derivative has extremum; see the insert in Fig. 3b and d), indicating a distinct water sorption mechanism and hence a different phase state of the particles before and after this RH point. Most likely, at low RH, D-glucose and AS / Gl (1:1) mixed particles are in the semi-solid amorphous state; the increase in humidity over glassy amorphous particles leads to a moisture-induced phase transition that occurs at a “glass transition relative humidity” of RHg≈48 % (Mikhailov et al., 2009).
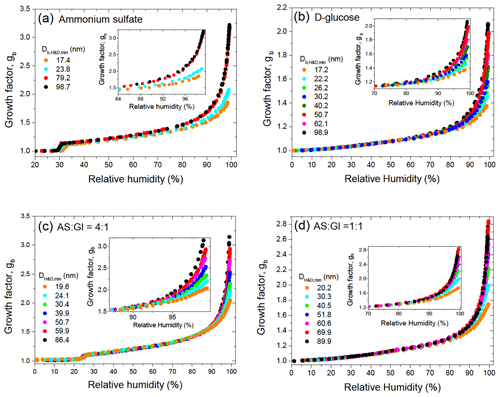
Figure 2Size-dependent growth factors, gb, of ammonium sulfate (a), D-glucose (b), and their mixture with mass ratios (c) and (d) obtained in the dehydration HHTDMA mode.
Continued water uptake converts the amorphous particles into concentrated solution droplets. Due to the very low molecular diffusivity of glasses, the uptake of water vapor by glassy aerosol particles is limited to surface adsorption, whereas above RHg the particles are in a viscous liquid state and absorb water in the particle bulk. Differences in water sorption mechanisms may explain why the growth factors of small particles at low RH are higher than for larger particles. At RH below RHg with the same water adsorption layer, the water film contribution to gb will be higher for small particles. For example, at a water layer thickness of 0.56 nm (i.e., two monolayers of water molecules), gb for aerosol particles of 19 and 100 nm are 1.031 and 1.006, respectively. This pattern is clearly seen for AC/Gl (1:1) particles with compact initial structures (i.e., Db,i=DH&D,min, and gb=gH&D) for which the growth factor of large particles at the same RH is lower than that for small particles (Fig. 1d). Note also that moisture-induced transformation from the semi-solid state to the solute state can occur gradually within a certain RH range (Mikhailov et al., 2009). All these features in the water uptake by amorphous particles at high concentrations (low growth factor values) lead to additional uncertainties when using the DKA method.
4.3 DKA-derived aw and as for single-component aqueous solution
The DKA-derived water activity for ammonium sulfate and D-glucose aerosol particles are shown in Fig. 4a and b, respectively. More information on the DKA calculation can be found in Sect. S1 in the Supplement. The estimated uncertainty of aw for pure species is shown in Fig. S2 in the Supplement. The aw values for ammonium sulfate (Fig. 4a) agree well with electrodynamic balance (EDB) measurements by Tang and Munkelwitz (1994), Clegg et al. (1995), and Chan et al. (1992) and are almost identical to the DKA-derived aw values previously reported by Cheng et al. (2015) and Extended Aerosol Inorganic Model (E-AIM III) of Wexler and Clegg (2002), including highly concentrated aqueous solutions. In general, the DKA-derived water activities obtained for pure ammonium sulfate particles reproduce the literature data well.
Figure 4b shows the DKA-derived aw values of D-glucose aerosol particles. Our data are consistent with the EDB measurements by Peng et al. (2001) up to XGl∼0.8. The observed divergence of the water activity at XGl>0.8 may be due to kinetic limitations in highly viscous semi-solid amorphous particles (Mikhailov et al., 2009), which prevail in EDB experiments with micrometer-sized particles. As noted in Sect. 4.2 and shown in Fig. 3d at %, particles undergo moisture-induced phase transition where semi-solid particles become concentrated solution droplets. This RH value is very close to aw∼49 %, below which a strong deviation between DKA- and EDB-derived aw is observed (Fig. 4b). If this is the case, the DKA-derived aw here is closer to the real aw.
At the same time, the DKA-derived aw values differ from those predicted by the AIOMFAC model (Zuend et al., 2008), with a maximum deviation of 6 % at XGl∼0.7. The discrepancy between measured and AIOMFAC-predicted aw values for carbohydrates has been noted previously and is attributed to relatively strong intramolecular interactions due to several polar groups in close proximity, not accounted for by the UNIFAC model within the AIOMFAC framework (Zuend et al., 2011).
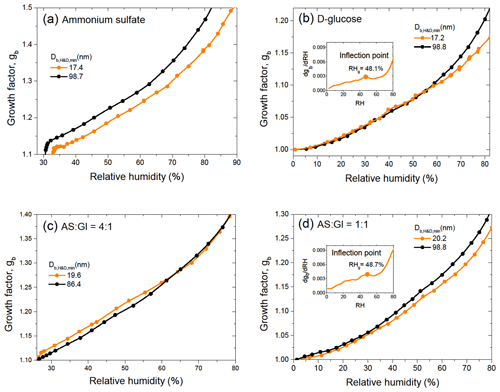
Figure 3Growth factor, gb, of ammonium sulfate (a), D-glucose (b), and their mixture with mass ratios (c) and (d) at low RH values for minimum and maximum sizes. The inserts (b and d) show derivative with inflection point RHg. The instrumental error in RHg does not exceed the symbol size.
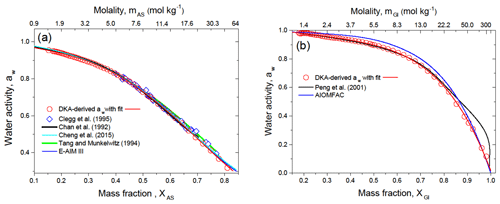
Figure 4DKA-derived water activity, aw, of ammonium sulfate (a) and D-glucose (b) aqueous solution at 298 K in comparison with literature data as a function of mass fraction and molality. The red line in panels (a) and (b) is a polynomial fit to all DKA data of aw. The best fit coefficients are listed in Table S1.
The multicomponent surface tension model of Li and Lu (2001) is based on the activity of the solute. To take advantage of this model, we calculated the activity coefficients of single solutes by numerical integration of Eq. (9) and then, using Eq. (7), their activities (see Sect. S2 in the Supplement for more details). The calculation results are shown in Fig. 5 in comparison with literature data. One can see that the ammonium sulfate activity is in good agreement with E-AIM (Fig. 5a) up to XAS=0.75 (mAS=22.7 mol kg−1). At higher XAS values, there is a discrepancy between our data and the model data. This discrepancy is most likely due to the uncertainty in DKA-derived aw in concentrated ammonium sulfate aqueous solutions (Fig. S2) and uncertainty in assessment of the integral Eq. (9) in the asymptotic region (ms→0) (Lakhanpal and Conway, 1960).
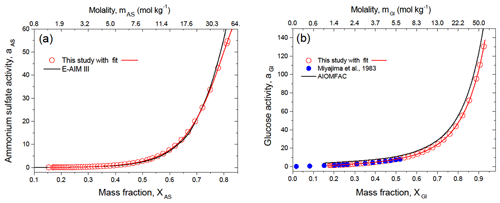
Figure 5Solute activity of ammonium sulfate (a) and D-glucose (b) in water at 298 K together with literature data as a function of mass fraction and molality. The red line in panels (a) and (b) are polynomial function with fitting parameters listed in Table S2.
Figure 5b shows the DKA-based activity of D-glucose. One can see that these values are in agreement with bulk measurements by Miyajima et al. (1983), obtained by the isopiestic method (blue symbols) available up to XGl=0.52 (mGl=6.0 mol kg−1). At the same time, a systematic deviation is observed between our results and the AIOMFAC model. As previously noted, the UNIFAC as a part of the AIOMFAC model does not match well with D-glucose and other carbohydrates, owing to strong intramolecular interactions of the polar groups in close proximity.
In general, the DKA-derived water activity and activity of ammonium sulfate and D-glucose in aqueous solutions reproduce the literature data well. For future applications, we fitted DKA-derived aw and as for single solutes with a polynomial function. The obtained fitting parameters are listed in Tables S1 and S2 in the Supplement.
4.4 Surface tension of the single solute solution droplet
Figure 6 shows the DKA-derived surface tension of ammonium sulfate and D-glucose aqueous solution droplets as a function of solute activity. For future use, we fit our data with the Li and Lu (2001) model (Eq. 12). The resulting values of and Ks together with the as fitting interval are listed in Table S3 in the Supplement. The negative values obtained for both ammonium sulfate and glucose solution droplets indicate that a negative adsorption occurs on the interface between the vapor and liquid phases, leading to surface tensions increasing with concentration, which is typical for surface-inactive compounds (Tuckermann, 2007; Aumann et al., 2010).
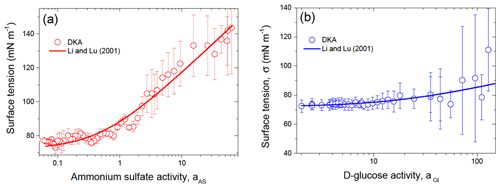
Figure 6Surface tension of ammonium sulfate (a) and D-glucose (b) aqueous solution droplets as a function of solute activity. The line in panels (a) and (b) is the model of Li and Lu (2001) (Eq. 12), with the best-fit parameters listed in Table S3.
For comparison with the literature data, in Fig. 7 we plotted the DKA surface tension in more accessible concentration scales, i.e., in mass fraction and molality. Figure 7a shows that the DKA-derived σ for ammonium sulfate solution is in agreement, within uncertainty, with the empirical approximation of Pruppacher and Klett (1997) and also consistent with the Dutcher et al. (2010) model up to XAS≈0.5 (mAS=7.6 mol kg−1). Above this value, the difference between DKA data and those by Dutcher et al. (2010) gradually increases with increasing concentration. This difference is due to the fact that model parameters used in Dutcher et al. (2010) (Sect. 3.3) are only valid for a concentration of ammonium sulfate not exceeding XAS=0.43 (mAS=5.69 mol kg−1), although the model structure allows for calculation of σ in the concentration range from dilute solution to molten salt (Eq. 11). Figure 7b shows the DKA-derived σ for D-glucose solution droplets as a function of mass fraction or molality together with the literature data. It can be seen that the DKA data are consistent with bulk measurements reported by Aumann et al. (2010). In the XGl range of 0.24 to 0.52, the average deviation between DKA-derived and literature σ is 2 %.
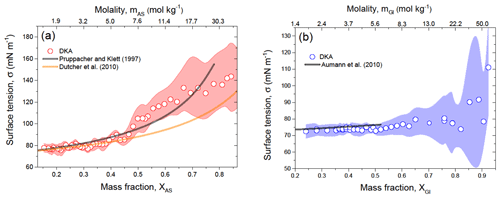
Figure 7DKA-derived surface tension of ammonium sulfate (a) and D-glucose (b) solution droplets as a function of mass fraction and molality in comparison with literature. The shaded area in panels (a) and (b) indicates uncertainty in the DKA retrieval σ.
Note that the uncertainty in the DKA-retrieval parameters is strongly influenced by of the accuracy of sw (or RH) and gb determination. At low gb (highly concentrated solutions) when is small, the uncertainty in the DKA-derived aw and σ increases with decreasing gb. In addition, phase state ambiguity and associated kinetic limitations can provide additional uncertainties. As an example, Fig. S3 in the Supplement shows experimental sw(Ds) dependences at low and high growth factors for D-glucose aerosol particles and corresponding uncertainties in the DKA-derived aw and σ.
4.5 DKA-derived aw and σ for mixed ammonium sulfate and D-glucose aerosol particles
In the absence of solute–solute interactions (“separate solutes”), the corresponding relationship for the water activity of the mixture is , where aw,i is the water activity of a pure aqueous solution of i at the same concentration as in the mixture (Mikhailov et al., 2004; Clegg and Seinfeld, 2006). Within this approach, referred to as “separate solute water activity” (SSWA), the water activity of mixed particles containing ammonium sulfate and D-glucose can be expressed as
Figure 8 shows the DKA-derived water activity of the mixed AS / Gl particles with a mass ratio of 4:1 (a) and 1:1 (b), as well as results of the AIOMFAC and SSWA models. One can see that the DKA-derived aw and both models are consistent up to Xs∼0.3. A further increase in concentration is accompanied by a significant deviation of the model aw values, both among themselves and from the DKA-derived aw. It appears that the SSWA approximation underestimates the specific interactions between ions and molecules in the droplet volume (higher aw), whereas the AIOMFAC model overestimates them (lower aw), especially in concentrated solutions.
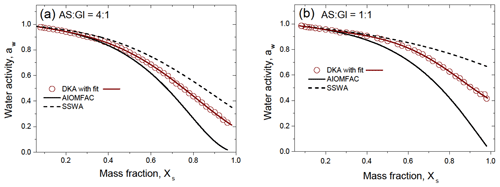
Figure 8Water activity of the aqueous solution of the mixed ammonium sulfate and D-glucose with mass ratio (a) and (b) compared to model data as a function of the mass fraction of the solute, Xs. The red line in panels (a) and (b) are polynomial function with fitting parameters listed in Table S1. SSWA denotes separate solute water activity (Eq. 17).
Figure 9 shows the DKA-retrieved surface tension of the mixed AS / Gl particles with mass ratios of 4:1 (a) and 1:1 (b), compared to the Li and Lu (2001) model. Both approaches, i.e., LiLu (1) and LiLu (2), do not match well with the HHTDMA–DKA-based σ. This discrepancy is especially pronounced for the mixed particles with an AS / Gl mass ratio of 1:1 (Fig. 9b). A slight lowering of the surface tension compared to that of water is already noticeable for a 4:1 mixture (Fig. 9a). The mixture (Fig. 9b) shows more significant surface tension depression, which is not a monotonic function of concentration. At Xs≈0.5, the σ reaches a minimum of 56.5 ± 3.0 mN m−1 (22 % reduction compared to pure water) and increases again with concentration. Further increase in surface tension with concentration is the result of solidification of aerosol particles. According to Eq. (10) at Xs→1, σ tends to the surface tension of molten state, σs, which for AS and Gl is 185 mN m−1 (Dutcher et al., 2010) and 150.9 mN m−1 (Docoslis et al., 2000), respectively. Thus, at Xs=1 the mole-fraction-weighted value of σ for and 1:1 is 179 and 170 mN m−1, respectively, which reasonably agrees with the DKA-derived σ at high Xs values (Fig. 9a and b).
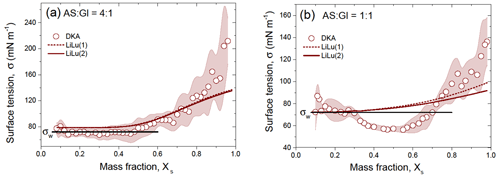
Figure 9DKA-derived surface tension of the aqueous solution of the mixed ammonium sulfate and D-glucose with mass ratio (a) and (b) together with Li and Lu (2001) model (LiLu (1) – Eq. (13) and LiLu (2) – Eq. (14)) as a function of mass fraction of the solute, Xs. The shaded area in panels (a) and (b) indicates the uncertainty in the DKA retrieval σ, the black line is the surface tension of pure water σw=72.0 mN m−1.
Note that the decrease in surface tension observed in Fig. 9b and its subsequent increase with concentration is reproduced in repeated measurements (see Fig. S4 in the Supplement). Most likely the reduction in surface tension at moderate concentrations is caused by the salting out effect (Setschenow, 1889; Kiss et al., 2005; Marcolli and Krieger, 2006; Frosch et al., 2011; Wang et al., 2014; Lin et al., 2020; Bzdek et al., 2020) and results from the interaction between ammonium sulfate ions and D-glucose molecules, facilitating the association of initially surface-inactive hydrophilic organic molecules into surface-active hydrophobic associates (quasi-macromolecules). Docoslis et al. (2000) analyzed the difference between polysaccharides (dextran, Ficoll) and their surface-inactive constituents (sucrose, glucose). They concluded that the contrasting results are caused by the differences in polar intermolecular reactivities of the monomeric and polymeric glucides. The singly dissolved monomeric sugars contribute strongly to the polar (Lewis acid–base) free energy of cohesion through the multiple interactions between their freely available electron donors and acceptors, through mutual interactions as well as through interactions with the surrounding water dipoles. However once covalently polymerized, these polysaccharides have lost the strong bipolarity of the monomeric sugar molecules. It can thus be assumed that, in an aqueous AS / Gl system, and ions effectively neutralize the bipolarity of the monomeric D-glucose molecules, facilitating their association into less-polar aggregates with reduced σ values, which are more readily accommodated at the air–droplet interface. Obviously, this mechanism is more pronounced for mixed particles (Fig. 9b) than for their 4:1 mixture (Fig. 9a), due to the higher D-glucose content (by a factor of 2.5).
In this study, the DKA method was applied to derive water activities and surface tension of pure ammonium sulfate and D-glucose as well as their mixtures with mass ratios of 4:1 and 1:1 based on shape-corrected hygroscopic growth factors for particles with diameter of 17–100 nm in the relative humidity range of 2.0 %–99.6 %. The obtained aw and σ values for pure ammonium sulfate and D-glucose droplet solution are in good agreement with the bulk measurements in the available concentration range for bulk methods. The DKA method was employed for the first time to determine aw and σ of AS / Gl mixed particles. Our data show that for dilute and moderate solution concentrations (Xs<0.3), the SSWA and AIOMFAC models are in agreement with the DKA-derived aw. However, the discrepancy between the model and our data increases rapidly with increasing solution concentration, with SSWA underestimating DKA data, while AIOMFAC overestimates it. Both ammonium sulfate and D-glucose are surface-inactive compounds with a positive slope, indicating negative solute adsorption. Interestingly, mixing them together leads to positive adsorption and reduction in surface tension at the air–surface interface (i.e., the AS / Gl mixture becomes a surface-active compound due to salting out). We suggest that AS ions neutralize polar groups of D-glucose, helping them to combine into less-polar aggregates with reduced σ values at the air–surface interface.
The error analysis showed that there are some factors affecting the accuracy of aw and σ determined by the DKA method. One of them is the irregular morphology of the initial nanoparticles, which leads to a size-dependent error in gb. Other factors are mainly characteristic of highly concentrated nanodroplet solutions, such as the large uncertainty of gb and ambiguity of the phase state. Both small growth factor changes at low RH and the different mechanisms of water uptake (surface adsorption vs. bulk absorption) lead to an increased uncertainty in the DKA-derived values of aw and σ. Thus, for D-glucose aerosol particles at RH near RHg=48 % and gb=1.08 (XGl=0.85; mGl=32.4 mol kg−1), the relative uncertainty in aw is 3.5 % and in σ it is 41.2 %, respectively.
As mentioned above, DKA assumes that thermodynamic parameters depend only on concentration and are independent of particle size. Good agreement of DKA-derived aw and σ with the literature data showed that, for particles in the size range of 20–100 nm, this approach is acceptable for single-component solutions of AS and Gl as well as their mixtures, at least at moderate concentrations. Additional HHTDMA–DKA studies are needed to evaluate the accuracy of this approximation for nanoparticles containing surface-active molecules. We plan to conduct such studies to compare the DKA-derived aw and σ with available experimental data and models that account for size-dependent bulk–surface partitioning.
The raw data used in this study are archived and are available on request by contacting the corresponding author, Alexei A. Kiselev (alexei.kiselev@kit.edu).
The supplement related to this article is available online at: https://doi.org/10.5194/acp-24-2971-2024-supplement.
EFM designed the study, performed the concomitant measurements, carried out the data analysis, and wrote the manuscript with input from all coauthors. SSV and AAK contributed to the discussion of the results.
The contact author has declared that none of the authors has any competing interests.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. While Copernicus Publications makes every effort to include appropriate place names, the final responsibility lies with the authors.
We would like to thank Ulrich Pöschl, Yafang Cheng, and Hang Su for their helpful recommendations that improved this paper.
The work of EFM and SSV was supported by the Russian Science Foundation, project 22-27-00258. AAK acknowledges financial support by the Helmholtz Association under the Atmosphere and Climate program (ATMO).
The article processing charges for this open-access
publication were covered by the Karlsruhe Institute of
Technology (KIT).
This paper was edited by Daniel Knopf and reviewed by two anonymous referees.
Anastasiadis, S. H., Chen, J. K., Koberstein, J. T., Siegel, A. F., Sohn, J. E., and Emerson, J. A.: The determination of interfacial tension by video image processing of pendant fluid drops, J. Colloid Interf. Sci., 119, 55–66, https://doi.org/10.1016/0021-9797(87)90244-X, 1987.
Andreae, M. O. and Rosenfeld, D.: Aerosol-cloud precipitation interactions. Part 1. The nature and sources of cloud-active aerosols, Earth-Sci. Rev., 89, 13–41, https://doi.org/10.1016/j.earscirev.2008.03.001, 2008.
Aumann, E., Hildemann, L. M., and Tabazadeh, A.: Measuring and modeling the composition and temperature-dependence of surface tension for organic solutions, Atmos. Environ., 44, 329–337, https://doi.org/10.1016/j.atmosenv.2009.10.033, 2010.
Bahadur, R. and Russell, L. M.: Effect of surface tension from MD simulations on size-dependent deliquescence of NaCl nanoparticles, Aerosol Sci. Tech., 42, 369–376, https://doi.org/10.1080/02786820802104965, 2008.
Biskos, G., Malinowski, A., Russell, L. M., Buseck, P. R., and Martin, S. T.: Nanosize effect on the deliquescence and the efflorescence of sodium chloride particles, Aerosol Sci. Tech., 40, 97–106, https://doi.org/10.1080/02786820500484396, 2006a.
Biskos, G., Russell, L. M., Buseck, P. R., and Martin, S. T.: Nanosize effect on the hygroscopic growth factor of aerosol particles, Geophys. Res. Lett., 33, L07801, https://doi.org/10.1029/2005GL025199, 2006b.
Bzdek, B. R., Reid, J. P., Malila, J., and Prisle, N. L.: The surface tension of surfactant-containing, finite volume droplets, P. Natl. Acad. Sci. USA, 117, 8335–8343, https://doi.org/10.1073/pnas.1915660117, 2020.
Chan, C. K., Flagan, R. C., and Seinfeld, J. H.: Water activity of solutions, Atmos. Environ. A-Gen., 26, 1661–1673, https://doi.org/10.1016/0960-1686(92)90065-S, 1992.
Cheng, Y., Su, H., Kopp, T., Mikhailov, E. F., and Pöschl, U.: Size dependence of phase transitions in aerosol nanoparticles, Nat. Commun., 6, 5923, https://doi.org/10.1038/ncomms6923, 2015.
Clegg, S. L. and Seinfeld, J. H.: Thermodynamic models of aqueous solutions containing inorganic electrolytes and dicarboxylic acids at 298.15 K. 1. The acids as nondissociating components, J. Phys. Chem. A, 110, 5692–5717, 2006.
Clegg, S. L., Ho, S. S., Chan, C. K., and Brimblecombe, P.: Thermodynamic properties of aqueous (NH4)2SO4 to high supersaturation as a function of temperature, J. Chem. Eng. Data, 40, 1079–1090, https://doi.org/10.1021/je00021a011, 1995.
Davies, J. F., Zuend, A., and Wilson, K. R.: Technical note: The role of evolving surface tension in the formation of cloud droplets, Atmos. Chem. Phys., 19, 2933–2946, https://doi.org/10.5194/acp-19-2933-2019, 2019.
Djikaev, Y. S., Bowles, R., Reiss, H., Hämeri, K., Laaksonen, A., and Väkevä, M.: Theory of size dependent deliquescence of nanoparticles: Relation to heterogeneous nucleation and comparison with experiments, J. Phys. Chem. B, 105, 7708–7722, https://doi.org/10.1021/jp010537e, 2001.
Docoslis, A., Giese, R. F., and van Oss, C. J.: Influence of the water–air interface on the apparent surface tension of aqueous solutions of hydrophilic solutes, Colloid. Surface. B, 19, 147–162, https://doi.org/10.1016/S0927-7765(00)00137-5, 2000.
Dutcher, C. S., Wexler, A. S., and Clegg, S. L.: Surface tensions of inorganic multicomponent aqueous electrolyte solutions and melts, J. Phys. Chem. A, 114, 12216–12230, https://doi.org/10.1021/jp105191z, 2010.
Forestieri, S. D., Staudt, S. M., Kuborn, T. M., Faber, K., Ruehl, C. R., Bertram, T. H., and Cappa, C. D.: Establishing the impact of model surfactants on cloud condensation nuclei activity of sea spray aerosol mimics, Atmos. Chem. Phys., 18, 10985–11005, https://doi.org/10.5194/acp-18-10985-2018, 2018.
Frosch, M., Prisle, N. L., Bilde, M., Varga, Z., and Kiss, G.: Joint effect of organic acids and inorganic salts on cloud droplet activation, Atmos. Chem. Phys., 11, 3895–3911, https://doi.org/10.5194/acp-11-3895-2011, 2011.
Gysel, M., Weingartner, E., Nyeki, S., Paulsen, D., Baltensperger, U., Galambos, I., and Kiss, G.: Hygroscopic properties of water-soluble matter and humic-like organics in atmospheric fine aerosol, Atmos. Chem. Phys., 4, 35–50, https://doi.org/10.5194/acp-4-35-2004, 2004.
Kiss, G., Tombácz, E., and Hansson, H.-C.: Surface tension effects of humic-like substances in aqueous extract of tropospheric fine aerosol, J. Atmos. Chem., 50, 279–294, https://doi.org/10.1007/s10874-005-5079-5, 2005.
Lakhanpal, M. L. and Conway, B. E.: A method of integration of the Gibbs–Duhem equation when activities of a solute are required from those of the solvent, Can. J. Chem., 38, 199–203, https://doi.org/10.1139/v60-027, 1960.
Lei, T., Su, H., Ma, N., Pöschl, U., Wiedensohler, A., and Cheng, Y.: Size-dependent hygroscopicity of levoglucosan and D-glucose aerosol nanoparticles, Atmos. Chem. Phys., 23, 4763–4774, https://doi.org/10.5194/acp-23-4763-2023, 2023.
Li, Z. B. and Lu, B. C. Y.: Surface tension of aqueous electrolyte solutions at high concentrations – representation and prediction, Chem. Eng. Sci., 56, 2879–2888, https://doi.org/10.1016/S0009-2509(00)00525-X, 2001.
Lin, J. J., Kristensen, T. B., Calderón, S. M., Malila, J., and Prisle, N. L.: Effects of surface tension time-evolution for CCN activation of a complex organic surfactant, Environ. Sci.-Proc. Imp., 22, 271–284, https://doi.org/10.1039/C9EM00426B, 2020.
Marcolli, C. and Krieger, U. K.: Phase changes during hygroscopic cycles of mixed organic/inorganic model systems of tropospheric aerosols, J. Chem. Phys., 110, 1881–1893, https://doi.org/10.1021/jp0556759, 2006.
Mikhailov, E., Vlasenko, S., Niessner, R., and Pöschl, U.: Interaction of aerosol particles composed of protein and saltswith water vapor: hygroscopic growth and microstructural rearrangement, Atmos. Chem. Phys., 4, 323–350, https://doi.org/10.5194/acp-4-323-2004, 2004.
Mikhailov, E., Vlasenko, S., Martin, S. T., Koop, T., and Pöschl, U.: Amorphous and crystalline aerosol particles interacting with water vapor: conceptual framework and experimental evidence for restructuring, phase transitions and kinetic limitations, Atmos. Chem. Phys., 9, 9491–9522, https://doi.org/10.5194/acp-9-9491-2009, 2009.
Mikhailov, E. F. and Vlasenko, S. S.: High-humidity tandem differential mobility analyzer for accurate determination of aerosol hygroscopic growth, microstructure, and activity coefficients over a wide range of relative humidity, Atmos. Meas. Tech., 13, 2035–2056, https://doi.org/10.5194/amt-13-2035-2020, 2020.
Mikhailov, E. F., Pöhlker, M. L., Reinmuth-Selzle, K., Vlasenko, S. S., Krüger, O. O., Fröhlich-Nowoisky, J., Pöhlker, C., Ivanova, O. A., Kiselev, A. A., Kremper, L. A., and Pöschl, U.: Water uptake of subpollen aerosol particles: hygroscopic growth, cloud condensation nuclei activation, and liquid–liquid phase separation, Atmos. Chem. Phys., 21, 6999–7022, https://doi.org/10.5194/acp-21-6999-2021, 2021.
Miyajima, K., Sawada, M., and Nakagaki, M.: Studies on aqueous solutions of saccharides. I. Activity coefficients of monosaccharides in aqueous solutions at 25 °C, B. Chem. Soc. Jpn., 56, 1620–1623, https://doi.org/10.1246/bcsj.56.1620, 1983.
Ovadnevaite, J., Zuend, A., Laaksonen, A., Sanchez, K. J., Roberts, G., Ceburnis, D., Decesari, S., Rinaldi, M., Hodas, N., Facchini, M. C., Seinfeld, J. H., and O'Dowd, C.: Surface tension prevails over solute effect in organic-influenced cloud droplet activation, Nature, 546, 637–641, https://doi.org/10.1038/nature22806, 2017.
Peng, C., Chow, A. H. L., and Chan, C. K.: Hygroscopic study of glucose, citric acid, and sorbitol using an electrodynamic balance: comparison with UNIFAC predictions, Aerosol Sci. Tech., 35, 753–758, https://doi.org/10.1080/02786820152546798, 2001.
Petters, S. S. and Petters, M. D.: Surfactant effect on cloud condensation nuclei for two-component internally mixed aerosols, J. Geophys. Res.-Atmos., 121, 1878–1895, https://doi.org/10.1002/2015JD024090, 2016.
Pöschl, U.: Atmospheric aerosols: composition, transformation, climate and health effects, Angew. Chem. Int. Edit., 44, 7520–7540, https://doi.org/10.1002/anie.200501122, 2005.
Prausnitz, J. M., Lichtenthaler, R. N., and de Azevedo, E. G.: Molecular Thermodynamics of Fluid-Phase Equilibria, Prentice Hall, Upper Saddle River, New Jersey, USA, 226, https://doi.org/10.1002/cjce.5450780222, 1999.
Prisle, N. L., Raatikainen, T., Laaksonen, A., and Bilde, M.: Surfactants in cloud droplet activation: mixed organic-inorganic particles, Atmos. Chem. Phys., 10, 5663–5683, https://doi.org/10.5194/acp-10-5663-2010, 2010.
Prisle, N. L., Dal Maso, M., and Kokkola, H.: A simple representation of surface active organic aerosol in cloud droplet formation, Atmos. Chem. Phys., 11, 4073–4083, https://doi.org/10.5194/acp-11-4073-2011, 2011.
Pruppacher, H. R. and Klett, J. D.: Microphysics of clouds and precipitation, Kluwer Academic Publishers, https://doi.org/10.1007/978-0-306-48100-0, 1997.
Robinson, R. A. and Stokes, R. H.: Electrolyte solutions, 2nd edn., Butterworths, London, ISBN 100408184906, 1970.
Ruehl, C. R., Davies, J. F., and Wilson, K. R.: An interfacial mechanism for cloud droplet formation on organic aerosols, Science, 351, 1447–1450, https://doi.org/10.1126/science.aad4889, 2016.
Schmedding, R. and Zuend, A.: A thermodynamic framework for bulk–surface partitioning in finite-volume mixed organic–inorganic aerosol particles and cloud droplets, Atmos. Chem. Phys., 23, 7741–7765, https://doi.org/10.5194/acp-23-7741-2023, 2023.
Setschenow, J.: Über die Konstitution der Salzlösungen auf Grund ihres Verhaltens zu Kohlensäure, Z. Phys. Chem., 4, 117–125, https://doi.org/10.1515/zpch-1889-0409, 1889.
Shapiro, E. L., Szprengiel, J., Sareen, N., Jen, C. N., Giordano, M. R., and McNeill, V. F.: Light-absorbing secondary organic material formed by glyoxal in aqueous aerosol mimics, Atmos. Chem. Phys., 9, 2289–2300, https://doi.org/10.5194/acp-9-2289-2009, 2009.
Swietlicki, E., Hansson, H.-C., Hämeri, K., Svenningsson, B., Massling, A., McFiggans, G., McMurry, P. H., Petäjä, T., Tunved, P., Gysel, M., Topping, D., Weingartner, E., Baltensperger, U., Rissler, J., Wiedensohler, A., and Kulmala, M.: Hygroscopic properties of submicrometer atmospheric aerosol particles measured with H-TDMA instruments in various environments – a review, Tellus B, 60, 432–469, https://doi.org/10.1111/j.1600-0889.2008.00350.x, 2008.
Tang, I. N. and Munkelwitz, H. R.: Water activities, densities, and refractive indices of aqueous sulfates and sodium nitrate droplets of atmospheric importance, J. Geophys. Res., 99, 18801–18808, https://doi.org/10.1029/94JD01345, 1994.
Tang, M., Chan, C. K., Li, Y. J., Su, H., Ma, Q., Wu, Z., Zhang, G., Wang, Z., Ge, M., Hu, M., He, H., and Wang, X.: A review of experimental techniques for aerosol hygroscopicity studies, Atmos. Chem. Phys., 19, 12631–12686, https://doi.org/10.5194/acp-19-12631-2019, 2019.
Topping, D. O., McFiggans, G. B., Kiss, G., Varga, Z., Facchini, M. C., Decesari, S., and Mircea, M.: Surface tensions of multi-component mixed inorganic/organic aqueous systems of atmospheric significance: measurements, model predictions and importance for cloud activation predictions, Atmos. Chem. Phys., 7, 2371–2398, https://doi.org/10.5194/acp-7-2371-2007, 2007.
Tuckermann, R.: Surface tension of aqueous solutions of water-soluble organic and inorganic compounds, Atmos. Environ., 41, 6265–6275, https://doi.org/10.1016/j.atmosenv.2007.03.051, 2007.
Wang, C., Lei, Y. D., Endo, S., and Wania, F.: Measuring and modeling the salting-out effect in ammonium sulfate solutions, Environ. Sci. Technol., 48, 22, 13238–13245, https://doi.org/10.1021/es5035602, 2014.
Wexler, A. S. and Clegg, S. L.: Atmospheric Aerosol Models for Systems Including the Ions H+, , Na+, , , Cl−, Br−, and H2O, J. Geophys. Res., 107, 4207, https://doi.org/10.1029/2001JD000451, 2002.
Zuend, A., Marcolli, C., Luo, B. P., and Peter, T.: A thermodynamic model of mixed organic-inorganic aerosols to predict activity coefficients, Atmos. Chem. Phys., 8, 4559–4593, https://doi.org/10.5194/acp-8-4559-2008, 2008.
Zuend, A., Marcolli, C., Booth, A. M., Lienhard, D. M., Soonsin, V., Krieger, U. K., Topping, D. O., McFiggans, G., Peter, T., and Seinfeld, J. H.: New and extended parameterization of the thermodynamic model AIOMFAC: calculation of activity coefficients for organic-inorganic mixtures containing carboxyl, hydroxyl, carbonyl, ether, ester, alkenyl, alkyl, and aromatic functional groups, Atmos. Chem. Phys., 11, 9155–9206, https://doi.org/10.5194/acp-11-9155-2011, 2011.