the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Measurement report: Age-dependent BVOC emissions in Eucalyptus urophylla: a comparison of leaf cuvette and branch chamber measurements
Xiao Tian
Jianqiang Zeng
Weihua Pang
Yuting Lu
Haofan Ran
Hao Guo
Zhaobin Mu
Wei Song
Xinming Wang
Biogenic volatile organic compound (BVOC) emission factors (Es) underpin air quality and climate models, yet current databases intermingle data from both greenhouse seedlings and field mature trees and from two enclosure techniques, leaf cuvettes and dynamic branch chambers, whose comparability has rarely been rigorously tested. Here we quantified BVOC emissions from Eucalyptus urophylla by pairing the two methods on a statistically representative number of 2-month-old seedlings in the laboratory and 2-year-old in-situ saplings measured at a managed plantations in subtropical China. Leaf-cuvette and branch-chamber determination of isoprene Es matched within 5 % for both age classes, demonstrating method equivalence. In contrast, tree age exerted a significant impact on both the magnitude and speciation of emissions. Seedlings emitted ∼ 50 % more isoprene and were enriched in cyclic monoterpenes like α-pinene and 1,8-cineole, whereas field-grown trees shifted toward highly reactive acyclic monoterpenes, with β-ocimenes accounted for over 85 % of the terpene flux and a double rise in sesquiterpenes. These ontogenetic shifts imply that one-third of the entries in global Es compilations, which are derived from seedling studies, are likely inappropriate as generic surrogates for natural forest emissions. Our results validate the use of either chamber type for measuring isoprene Es, highlight the need for improved analytical sensitivity before extending this equivalence to terpenes, and call for systematic, large-sample, branch-level measurements of adult trees to produce representative Es values. Incorporating age-resolved emission factors into models will refine estimates of ozone and secondary organic aerosol formation in fast-growing subtropical plantations and other managed forests worldwide.
- Article
(612 KB) - Full-text XML
-
Supplement
(979 KB) - BibTeX
- EndNote
Terrestrial vegetation release on the order of ∼ 1000 Tg yr−1 for biogenic volatile organic compounds (BVOCs), representing ∼ 90 % of global non-methane VOCs injected to the troposphere (Guenther et al., 1995, 2012). Owing to their high reactivity with atmospheric lifetimes of only minutes to hours (Atkinson and Arey, 2003), BVOCs can strongly modulate the oxidative capacity of the troposphere and drive the production of ozone and secondary organic aerosols (Di Carlo et al., 2004; Peñuelas and Staudt, 2010; Scott et al., 2017; Pfannerstill et al., 2024; Weber et al., 2024). Reliable quantification of these fluxes is therefore essential for assessing their impacts on air quality and climate.
Current bottom-up BVOC emission inventories rely on species-specific emission factors (Es) that are applied within models such as the Model of Emissions of Gases and Aerosols from Nature (MEGAN; Guenther et al., 2012, 2020). Inaccurate emission factors are now recognized as a leading source of uncertainty in regional and global estimates of BVOC budgets (Guenther et al., 2012; Zhang et al., 2020; Wang et al., 2023). Decades of enclosure studies produced several widely used databases, like the Sheffield (Hewitt and Street, 1992), UCAR (Wiedinmyer et al., 2004), and most recently, the tropical plant compilation of Mu et al. (2022). However, two methodological and biological issues remain unresolved.
Both leaf cuvettes and dynamic branch chambers have been used for measuring Es. Guenther et al. (1994) suggested, from a literature survey, that isoprene Es with leaf cuvette are approximately 75 % higher than branch-based values, but a rigorous, side-by-side validation on the same trees is still lacking. Whether the two techniques can be used interchangeably is therefore uncertain.
As for the tree age, approximately one-third of database entries originate from greenhouse or growth-chamber seedlings (Guenther, 2013), yet seedling's physiology differs markedly from that of mature trees. Limited case studies on Eucalyptus spp. point to pronounced ontogenetic shifts in both the magnitude and speciation of BVOC emissions (Street et al., 1997; He et al., 2000; Winters et al., 2009). Meanwhile, considerable uncertainty may result from the potential intraspecific variability and chemo-diversity of BVOC emissions (Loreto et al., 2009; Staudt and Visnadi, 2023; Zeng et al., 2024). Previous investigations, however, employed small sample sizes and heterogenous protocols, leaving the statistical representativeness of age effects largely unconstrained.
Addressing these gaps requires large-sample, method-controlled measurements that span contrasting developmental stages. Here we report parallel cuvette and branch chamber determination of BVOC emissions from Eucalyptus urophylla seedings grown under laboratory conditions (2-month-old) and saplings (2-year-old) measured in situ at a managed plantation in subtropical China. The objectives are to (1) quantitatively test the equivalence of the two chamber types for isoprene and, where detection limits permit, for terpenes; (2) disentangle how tree age influences both emission factors and chemical composition; and (3) assess the implications for constructing representative Es databases and for simulating ozone and secondary organic aerosol formation in rapidly expanding plantation forests. By combining method inter-comparison with statistically robust sampling across ontogeny, our work provides critical benchmark for future BVOC inventories and modeling frameworks. It should be noted that we only focus on the seedling to sapling transition, using 2-month-old seedlings and 2-year-old field grown saplings as contrasting stages; we do not attempt to represent fully mature trees.
2.1 Study sites and plant materials
Laboratory measurements were performed at the Guangzhou Institute of Geochemistry (GIG, 23.145° N, 113.364° E). Leaf experiments were conducted on 1–3 June 2023, and branch-chamber measurements were conducted on 10–13 June 2023 (7 d separation). Field measurements were carried out in a managed E. urophylla plantation at Heshan (22.649° N, 112.904° E), Guangdong province, China. Leaf and branch measurements were performed concurrently on the same days during two campaigns: 11–13 July and 26–31 July 2022. The study region experiences a humid subtropical monsoon climate, with a 30-year mean temperature of 22 °C and annual precipitation of ∼ 1700 mm (Mu et al., 2023; Zeng et al., 2024). As shown in Table 1, two age classes, 2-month-old and 2-year-old, were investigated, with seedlings (15 for branch chamber and 50 for leaf cuvette) measured in laboratory and 2-year-old saplings (26 for branch chamber and 114 for leaf cuvette) measured in situ at the plantation. Seedlings were purchased from a local nursery and measured by both leaf cuvette and dynamic branch chamber. These trees were placed in an open area of GIG and acclimated to the ambient condition for two weeks before measurements; no greenhouse or climate-chamber but outdoor conditions were used. Field trees for leaf cuvette were randomly chosen from > 8 ha of homogeneous plantation to ensure spatial representativeness, while sunlit, single-layer branches were selectively chosen for dynamic chamber measurements. Both seedlings and saplings were sourced from the same clonal line and exhibited a uniform terpene chemotype, minimizing genotype/chemotype variability. The clonal identity of all seedlings and saplings was confirmed using nursery propagation records and plantation establishment documents. This is also supported by the similar monoterpene composition between stressless saplings and seedlings (detailed discussions see Sect. 3.2.3). All measurements, both in the laboratory and in the field, were conducted between 09:00 and 17:00 local time under sunny conditions, with 33 %, 56 %, 10 % of the samples collected in the morning (09:00–12:00), midday (12:00–15:00), and late afternoon (15:00–17:00), respectively, in the field. The real-world (non-normalized) emission rates of BVOCs by time-of-day bins was provided in Fig. S1. Midday maximum PAR exceeded 1200 µmol m−2 s−1 during both campaigns (Figs. S2 and S3). Mean daily air temperatures during the campaigns were 31.4 °C (seedling campaign at GIG) and 30.3 °C (Field campaign; Fig. S4).
2.2 Enclosure measurements
2.2.1 Leaf cuvette
Leaf cuvette fluxes were obtained with a LI-6800 portable photosynthesis system (LI-COR, Lincoln, NE, USA) fitted with a 6800-01A fluorometer head (aperture 6 cm2, air flow 500 µmol s−1). We imposed standard conditions for emission factors (Es): leaf temperature 30 °C, PAR 1000 µmol m−2 s−1, CO2 400 µmol mol−1, and 55 % RH. The LI-6800 maintains closed loop control and continuously records actual leaf temperature and PAR. Because measurements were made at the standard reference conditions, cuvette fluxes equal Es after correction to leaf dry mass. After 5 min stabilization of photosynthesis, 200 mL min−1 of outlet air was diverted for 2 min through Tenax TA/Carbograph 5TD adsorbent cartridges (Markes International Ltd, Bridgend, UK) using a dual-channel pump (ZC-QL, Zhejiang Hengda Instrumentation Ltd., Zhejiang, China). Detailed leaf handling, leaf area and dry mass determination are provided in Section S1 and are also described in Zeng et al. (2024, 2025a, b).
2.2.2 Dynamic branch chamber
A cylindrical PMMA chamber (∅ 25 cm × 28 cm, 13.7 L) internally coated with FEP film was used for branch-level measurements. The charcoal- and KI-scrubbed ambient air (9 L min−1) was supplied by a mass-flow controller (Alicat Scientific, Inc., Tucson, AZ, USA) coupled with an oil-free pump (MPU2134-N920-2.08; KNF, Freiburg, Germany), then it was well mixed with PTFE-bladed fans in the chamber (Zeng et al., 2022a, 2025c). For comparability with leaf cuvette measurements, sunlit branches (3–5 m above ground) with ∼ 6–10 leaves were selected, avoiding mutual overlap so that foliage formed a single layer. Branches showing visible self-shading were not sampled. These practices follow our goal of minimizing light heterogeneity within the chamber. When the selected sunlit branches were enclosed; fluxes were allowed to stabilize for 1–2 h before sampling. Outlet air (and inlet blanks) was drawn at 200 mL min−1 for 10 min by an automatic sampler (JEC921; Jectec Science and Technology, Co., Ltd, Beijing, China) onto the same adsorbent tubes as above. The chromatograms of a representative BVOC sample and its corresponding blank sample of inlet air during the field study was shown in Fig. S5. Concurrent meteorological and radiometric variables were logged continuously (Rotronic HC2A-S RH/T probes; LI-1500 PAR sensor; OMEGA/RKC thermocouples). Specifically, leaf temperature was measured by thermocouples attached on the leaf, while PAR was measured by a LI-1500 PAR sensor placed on the top of the chamber, these parameters were used to calculate the standard emission factors (Es). More details about the branch sampling are provided in Section S2 and Zeng et al. (2022a).
2.3 Thermal desorption-GC/MS analysis
Tubes were analyzed within 7 d with a TD-100 system (Markes) coupled to an Agilent 7890 GC-5975 MSD. Primary cartridge desorption was 280 °C, cold-trapping at −10 °C, then desorption at 320 °C. Separation employed an HP-5 MS (30 m × 0.25 mm × 0.25 µm) column. The GC oven temperature program was started at 35 °C (3 min), 5 °C min−1 to 100 °C (1 min), 10 °C min−1 to 120 °C (12 min), and 20 °C min−1 to 260 °C (2 min). Identification used authentic standards, while quantification based on calibration curves. Figure S6 shows the calibration curve for β-ocimene (cis- and trans-) as an example. Ocimene-like compounds such as 3,6-dimethyl-1,3,7-octatriene, 3,4-dimethyl-2,4,6-octatriene, and alloocimene were identified by the NIST library and quantified based on β-ocimene. More information about the analysis and quantification of other MTs are given in Section S3 and Zeng et al. (2022a, b).
2.4 Calculation of emission rates and emission factors
For branch data, emission rates (E, µg g−1 h−1) were calculated as
where F is chamber flow (L h−1) and gdw the dry leaf mass. Branch chamber fluxes measured under ambient conditions were standardized to 30 °C and 1000 µmol m−2 s−1 using the MEGAN/Guenther temperature and light response functions with the measured leaf temperature and PAR, yielding Es directly comparable to cuvette derived values. The temperature- and light-dependent function was applied for light dependent compounds, while a temperature-only function was used for light-independent monoterpenes (β = 0.10 K−1) and sesquiterpenes (β = 0.17 K−1). More details about the calculation of emission factors are provided in Sect. S4 and in Zeng et al. (2023).
Notably, Es can be normalized either to leaf dry mass ( µg g−1 h−1) or to projected leaf area ( µg m−2 h−1). Because leaf mass per area (LMA, g m−2) increases with age in E. urophylla (Table 1), the two normalizations emphasize different aspects of physiology and scaling (Es-area = Es-mass × LMA). To remove duplication and prevent unit confusion, we only report mass-based Es in the main text and present all area-based values in the Supplement.
2.5 Statistical analysis
All statistics were performed in Excel 2019. Isoprene and terpene Es were log-normal, and they were log-transformed before testing. Equality of means between age classes or methodologies was evaluated with Welch's t-test; distributions that failed Shapiro-Wilk normality (p<0.05) were compared with Mann-Whitney U. Significance was accepted at p<0.05. Results are reported as mean ±1 SD unless otherwise specified.
3.1 Method inter-comparison for isoprene Es
Side-by-side measurements of E. urophylla seedlings (2 months old) and 2-years-old trees showed that the two dominant enclosure techniques yielded statistically indistinguishable isoprene Es. As shown in Fig. 1, for seedlings measured at GIG, cuvette-derived isoprene Es averaged at 102.4 ± 34.1 µg g−1 h−1 (N=50) versus 107.7 ± 34.9 µg g−1 h−1 from branch chambers (N=15), a non-significant 5 % difference (p=0.61). In the field, 2-year-old trees exhibited similarly close agreement: 66.9 ± 31.4 µg g−1 h−1 (N=114) from leaf cuvettes and 69.8 ± 21.2 µg g−1 h−1 (N=26) from branch chambers (p=0.57). A direct test in which the same branch was first sub-sampled leaf-by-leaf and then enclosed intact confirmed parity within analytical uncertainty (Fig. S7).
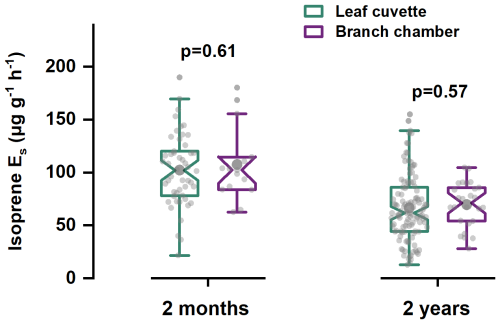
Figure 1Comparison of isoprene Es from leaf cuvettes with those from dynamic branch chambers for both 2-month-old and 2-year-old trees.
Leaf cuvette and dynamic branch chamber are the two most widely employed techniques for measuring plant BVOC emissions (Niinemets et al., 2011). The present study provides the first large-sample validation that leaf cuvette and dynamic branch chamber protocols are interchangeable for isoprene, contradicting the ∼ 75 % bias inferred from the earlier literature meta-analysis (Guenther et al., 1994). The convergence arises despite distinct air flow regimes (0.75 L min−1 vs. 9 L min−1) and path lengths, implying that adsorptive/ozonolysis losses are negligible for this highly volatile compound. Because branch chambers integrate six to ten leaves per branch, their representativeness is at least as good as that of cuvettes as previously demonstrated for Eucalyptus by Zeng et al. (2024), and they remain advantageous when mature crowns are inaccessible.
Establishing methodological equivalence for isoprene enables age effects to be probed with confidence using the more logistically efficient branch chamber dataset (Sect. 3.2). Whether the same holds for lower-volatility monoterpenes and sesquiterpenes remains an open question due to the assessment of method equivalence was precluded by their lower fluxes and the limited air volumes sampled. Because terpene emissions are more sensitive to wall losses and analytical detection limits than isoprene, systematic inter-comparison studies with larger sample volumes and a broader range of plant species are still required.
Moreover, the equivalence we observed for isoprene was obtained under sunlit, low LAI branches (∼ 6–10 leaves) chosen to limit mutual shading. Larger or denser branches that capture multi-layer foliage can depress within chamber PAR for a subset of leaves and thus bias light dependent fluxes low relative to single leaf cuvettes; in such cases, method differences may not remain negligible. We therefore recommend selecting unshaded branches, documenting within enclosure PAR (or its ratio to ambient), and reporting the number of enclosed leaves and leaf layering as part of QA/QC.
3.2 Ontogenetic controls on the magnitude and speciation of BVOC emissions
3.2.1 Speciation diversity
Dynamic branch chamber measurements revealed 12 BVOC species in 2-month-old seedlings (isoprene, 8 monoterpenes, 3 sesquiterpenes; Fig. S8) versus 17 species in 2-year-old trees (isoprene, 11 monoterpenes, 5 sesquiterpenes; Table S1). Thus, chemical richness increased by ∼ 40 % with age, consistent with the view that metabolic complexity develops as trees mature (Satake et al., 2024).
3.2.2 Emission magnitude
Isoprene dominated the flux from both age classes but its standardized Es declined significantly (p<0.001) from 107.7 ± 34.9 µg g−1 h−1 in seedlings to 69.8 ± 21.2 µg g−1 h−1 in two-year-old trees. When normalized by leaf area, however, the two groups were indistinguishable (Fig. S9a), indicating that the mass-based contrast mainly reflects an increase in leaf mass per area (LMA) with tree age (Fig. S10). Seedlings therefore allocated a larger fraction of assimilated carbon to isoprene (Fig. S9c) despite exhibiting lower net photosynthesis (Pn, Fig. S9b), suggesting a typical growth-defense trade-off of early ontogeny. Consistent with our findings, isoprene Es for the 1-year-old E. globulus was 5-fold higher than that for the 7-year-old individual (Street et al., 1997). Winters et al. (2009) also documented lower isoprene Es in four 10-year-old eucalyptus species compared to their seedlings measured by He et al. (2000).
3.2.3 Terpene speciation shift
Total monoterpene emissions rose nearly 6-fold with tree age (1.09 vs. 6.14 μg g−1 h−1), but the increase was almost entirely due to acyclic β-ocimenes, whose Es leapt from 0.133 ± 0.06 to 5.33 ± 4.61 µg g−1 h−1 (Fig. 2 and Table S1). By contrast, Es for cyclic α-pinene, limonene, and 1,8-cineole declined by 30 %–65 % (Fig. 2). Consequently, terpene composition shifted from 75 % cyclic (seedlings) to 85 % acyclic (2-year-old trees) (Fig. 2). Sesquiterpenes doubled in absolute terms but remained a minor (< 1 %) proportion of total BVOCs.
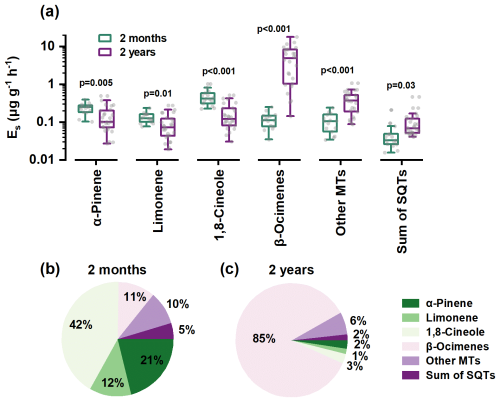
Figure 2Comparison of emission factors (a) and compositions (b, c) between 2-month-old and 2-year-old trees.
During the branch experiments, both seedlings and saplings were exposed to high growth temperature conditions, which could lead to significant stress on plant physiology as discussed by Zeng et al. (2025d). This was evidenced by the substantial emissions of typical stress-induced β-ocimene from saplings. However, although seedlings were also subjected to heat stress, they did not exhibit similarly high β-ocimene emissions but dominated by α-pinene and 1,8-cineole, closely resembling those under non-stress saplings measured during the warm dry season (Fig. S11). This indicates that stress-induced gene expression may require time to accumulate, and that seedlings grown in greenhouses, having not experienced environmental stresses, may not adequately represent the emission characteristics of trees in natural field conditions.
Such a cyclic-to-acyclic transition has been observed in other Eucalyputus species and might be attributed to age-dependent expression of terpene synthesis and to selectively pressures from biotic/abiotic stress in the field (Monson et al., 2021; Pollastri et al., 2021). For example, most previous studies reported that cyclic α-pinene and 1,8-cineole were the dominant MTs in stressless lab-grown seedlings (Evans et al., 1982; Guenther et al., 1991; He et al., 2000; Tsui et al., 2009; Malik et al., 2019), whereas acyclic MTs like β-ocimenes were generally low or even undetectable. In contrast, most stress-rich field-grown trees could emit large amounts of acyclic β-ocimenes (Street et al., 1997; Nunes and Pio, 2001; Sørensen et al., 2020; Purser et al., 2020, 2021; Nagalingam et al., 2023).
Both isoprene and β-ocimenes are known to play key roles in plant antioxidant defense. Isoprene helps scavenge reactive oxygen species (ROS) (Jardine et al., 2014), maintain membrane stability (Sharkey and Singsaas, 1995; Pollastri et al., 2019, 2021), and regulate antioxidative processes (Zuo et al., 2019; Monson et al., 2021), whereas the highly reactive β-ocimene may act as an even more efficient, direct antioxidant against ROS. The highly reactive β-ocimenes can quench ROS more efficiently than isoprene (Pollastri et al., 2021), providing a plausible advantage for field-grown trees exposed to stronger light, heat, drought, and herbivory.
Environmental factors, particularly growth temperature, light availability, and soil moisture/nutrients, can modulate BVOC emissions (Monson et al., 1994; Harley et al., 1994, 1996, 1997; Fall and Wildermuth, 1998; Funk et al., 2006; Guenther et al., 2006, 2012; Yuan et al., 2020). In our study, both campaigns were conducted under clear-sky, peak-summer conditions with comparable ambient temperature and high midday PAR (Figs. S1–3). A simple sensitivity using the MEGAN/Guenther acclimation scheme (Guenther et al., 2012) indicates that, a ∼ 10 % change in growth PAR would change standardized Es by ∼ 8 %, and a +1.1 °C change in growth temperature by ∼ 11 %. These effects are small relative to the age-related differences reported here, such as ∼ 6-fold increase in total monoterpenes and a > 40-fold rise in β ocimenes from seedlings to saplings (Fig. 2 and Table S1). Nevertheless, because we did not measure soil nutrient or moisture status, parallel experiments under controlled and co-located growth conditions are warranted to fully disentangle ontogeny from environment.
It should be noted that the seedlings used here were cultivated trees, where soil properties could be parallel with those in previous seedling-based studies. In contrast, the taller trees measured under field conditions reflect realistic and natural growth conditions. The substantial differences in Es and emission composition between seedlings and saplings underscore that seedling measurements are inappropriate as generic surrogates for natural forest emissions. Despite this, all age contrasts presented here are seedlings vs. saplings; extrapolation to mature trees requires additional in-situ measurements.
Notably, we counted the most complete Es database currently used by the MEGAN model (Wiedinmyer et al., 2004; Guenther et al., 2012), showing that one-third of the publications in this database were based on greenhouse seedlings. Our results show that these data tend to overestimate canopy-scale isoprene fluxes of subtropical eucalyptus plantation and under-represent atmospheric reactivity by excluding large β-ocimene emissions from these tree canopies. Incorporating age-resolved Es and prioritizing statistically robust and in-situ sampling of branches from adult trees, particularly in rapidly expanding subtropical plantations, will reduce inventory uncertainty and refine predictions of ozone and secondary organic aerosol formation.
Parallel measurements on E. urophylla seedlings (2 months) and saplings (2 years) show that leaf cuvettes and dynamic branch chambers yield indistinguishable isoprene Es when applied to sunlit, single-layer branches. Age exerts a strong control on speciation: seedlings have higher mass-based isoprene and are enriched in cyclic monoterpenes, whereas saplings exhibit ∼ 6-fold higher total monoterpenes dominated by β-ocimenes and increased sesquiterpenes. These findings indicate that laboratory-grown seedlings are not reliable proxies for field-grown saplings, and likely not for mature trees either. Despite this, other drivers, particularly soil properties (e.g., nitrogen availability, moisture) were not resolved here and may also influence emissions. This therefore motivates age-stratified and in-situ branch measurements on mature trees under parallel growth conditions to comprehensively probe age effects. Future studies should extend such measurements to other high isoprene genera (e.g. Quercus, Populus) and to tropical species that dominate global BVOC budgets, evaluate method comparability for low-volatility terpenes using larger-volume or adsorption-minimized chambers, and couple physiological measurements with transcriptomics to unravel the molecular basis of the observed metabolic shift from cyclic to acyclic terpenes.
The measurement data used in this study can be available at https://doi.org/10.17632/jw8g8gkm5t.1 (Zeng, 2025).
The supplement related to this article is available online at https://doi.org/10.5194/acp-26-6213-2026-supplement.
JZ and XT designed and carried out the experiments with the support of WP, YL, HR, ZM, HG, and WS. JZ and XT analyzed the samples in the lab. JZ and XT analyzed the data and prepared the original manuscript. XW and YZ revised the manuscript.
The contact author has declared that none of the authors has any competing interests.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. The authors bear the ultimate responsibility for providing appropriate place names. Views expressed in the text are those of the authors and do not necessarily reflect the views of the publisher.
This work is supported by the National Natural Science Foundation of China (42321003 and 42507150), the National Key Research and Development Program (2024YFC3714300), the Postdoctoral Fellowship Program of CPSF (GZB20250107), the Department of Science and Technology of Guangdong (2023B0303000007).
This research has been supported by the National Natural Science Foundation of China (42321003 and 42507150), the National Key Research and Development Program of China (2024YFC3714300), the Postdoctoral Fellowship Program of CPSF (GZB20250107), the Department of Science and Technology of Guangdong (grant no. 2023B0303000007).
This paper was edited by Kelley Barsanti and reviewed by three anonymous referees.
Atkinson, R. and Arey, J.: Gas-phase tropospheric chemistry of biogenic volatile organic compounds: a review, Atmos. Environ., 37, S197–S219, https://doi.org/10.1016/s1352-2310(03)00391-1, 2003.
Di Carlo, P., Brune, W. H., Martinez, M., Harder, H., Lesher, R., Ren, X. R., Thornberry, T., Carroll, M. A., Young, V., Shepson, P. B., Riemer, D., Apel, E., and Campbell, C.: Missing OH reactivity in a forest: Evidence for unknown reactive biogenic VOCs, Science, 304, 722–725, https://doi.org/10.1126/science.1094392, 2004.
Evans, R. C., Tingey, D. T., Gumpertz, M. L., and Burns, W. F.: Estimates of isoprene and monoterpene emission rates in plants, Bot. Gaz., 143, 304–310, https://doi.org/10.1086/botanicalgazette.143.3.2474826, 1982.
Fall, R. and Wildermuth, M. C.: Isoprene synthase: From biochemical mechanism to emission algorithm, J. Geophys. Res., 103, 25599–25609, https://doi.org/10.1029/98jd00808, 1998.
Funk, J. L., Giardina, C. P., Knohl, A., and Lerdau, M. T.: Influence of nutrient availability, stand age, and canopy structure on isoprene flux in a Eucalyptus saligna experimental forest, J. Geophys. Res., 111, G02012, https://doi.org/10.1029/2005jg000085, 2006.
Guenther, A., Hewitt, C. N., Erickson, D., Fall, R., Geron, C., Graedel, T., Harley, P., Klinger, L., Lerdau, M., McKay, W. A., Pierce, T., Scholes, B., Steinbrecher, R., Tallamraju, R., Taylor, J., and Zimmerman, P.: A global model of natural volatile organic compound emissions, J. Geophys. Res., 100, 8873–8892, https://doi.org/10.1029/94jd02950, 1995.
Guenther, A., Karl, T., Harley, P., Wiedinmyer, C., Palmer, P. I., and Geron, C.: Estimates of global terrestrial isoprene emissions using MEGAN (Model of Emissions of Gases and Aerosols from Nature), Atmos. Chem. Phys., 6, 3181–3210, https://doi.org/10.5194/acp-6-3181-2006, 2006.
Guenther, A. B., Jiang, X., Heald, C. L., Sakulyanontvittaya, T., Duhl, T., Emmons, L. K., and Wang, X.: The Model of Emissions of Gases and Aerosols from Nature version 2.1 (MEGAN2.1): an extended and updated framework for modeling biogenic emissions, Geosci. Model Dev., 5, 1471–1492, https://doi.org/10.5194/gmd-5-1471-2012, 2012.
Guenther, A. B., Jiang, X., Shah, T., Huang, L., Kemball-Cook, S., and Yarwood, G.: Model of emissions of gases and aerosol from nature version 3 (MEGAN3) for estimating biogenic emissions, Springer Proceedings in Complexity, in: Air pollution modeling and its application XXVI, edited by: Mensink, C., Gong, W., and Hakami, A., 187–192, Springer International Publishing, https://doi.org/10.1007/978-3-030-22055-6_29, 2020.
Guenther, A.: Biological and chemical diversity of biogenic volatile organic emissions into the atmosphere, ISRN Atmos. Sci., 2013, 1–27, https://doi.org/10.1155/2013/786290, 2013.
Guenther, A., Zimmerman, P., and Wildermuth, M.: Natural volatile organic compound emission rate estimates for United States woodland landscapes, Atmos. Environ., 28, 1197–1210, https://doi.org/10.1016/1352-2310(94)90297-6, 1994.
Guenther, A. B., Monson, R. K., and Fall, R.: Isoprene and monoterpene emission variability: Observations with Eucalyptus and emission rate algorithm development, J. Geophys. Res.-Atmos., 96, 10799–10808, https://doi.org/10.1029/91jd00960, 1991.
He, C. R., Murray, F., and Lyons, T.: Monoterpene and isoprene emissions from 15 Eucalyptus species in Australia, Atmos. Environ., 34, 645–655, https://doi.org/10.1016/s1352-2310(99)00219-8, 2000.
Hewitt, C. N. and Street, R. A.: A qualitative assessment of the emission of nonmethane hydrocarbon compounds from the biosphere to the atmosphere in the UK: present knowledge and uncertainties, Atmos. Environ. A, 26, 3069–3077, 1992.
Harley, P. C., Litvak, M. E., Sharkey, T. D., and Monson, R. K.: Isoprene emission from Velvet bean leaves – Interactions among nitrogen availability, growth photo flux density, and leaf development, Plant Physiol., 105, 279–285, https://doi.org/10.1104/pp.105.1.279, 1994.
Harley, P., Guenther, A., and Zimmerman, P.: Effects of light, temperature and canopy position on net photosynthesis and isoprene emission from sweetgum (Liquidambar styraciflua) leaves, Tree Physiol., 16, 25–32, https://doi.org/10.1093/treephys/16.1-2.25, 1996.
Harley, P., Guenther, A., and Zimmerman, P.: Environmental controls over isoprene emission in deciduous oak canopies, Tree Physiol., 17, 705–714, https://doi.org/10.1093/treephys/17.11.705, 1997.
Jardine, K., Chambers, J., Alves, E. G., Teixeira, A., Garcia, S., Holm, J., Higuchi, N., Manzi, A., Abrell, L., Fuentes, J. D., Nielsen, L. K., Torn, M. S., and Vickers, C. E.: Dynamic balancing of isoprene carbon sources reflects photosynthetic and photorespiratory responses to temperature stress, Plant Physiol., 166, 2051–2064, https://doi.org/10.1104/pp.114.247494, 2014.
Loreto, F., Bagnoli, F., and Fineschi, S.: One species, many terpenes: matching chemical and biological diversity, Trends Plant Sci., 14, 416–420, https://doi.org/10.1016/j.tplants.2009.06.003, 2009.
Malik, T. G., Gajbhiye, T., and Pandey, S. K.: Some insights into composition and monoterpene emission rates from selected dominant tropical tree species of Central India: Plant-specific seasonal variations, Ecol. Res., 34, 821–834, https://doi.org/10.1111/1440-1703.12058, 2019.
Monson, R. K., Harley, P. C., Litvak, M. E., Wildermuth, M., Guenther, A. B., Zimmerman, P. R., and Fall, R.: Environmental and developmental controls over the seasonal pattern of isoprene emission from aspen leaves, Oecologia, 99, 260–270, https://doi.org/10.1007/bf00627738, 1994.
Monson, R. K., Weraduwage, S. M., Rosenkranz, M., Schnitzler, J. P., and Sharkey, T. D.: Leaf isoprene emission as a trait that mediates the growth-defense tradeoff in the face of climate stress, Oecologia, 197, 885–902, https://doi.org/10.1007/s00442-020-04813-7, 2021.
Mu, Z., Llusià, J., Zeng, J., Zhang, Y., Asensio, D., Yang, K., Yi, Z., Wang, X., and Peñuelas, J.: An overview of the isoprenoid emissions from tropical plant species, Front. Plant Sci., 13, 833030, https://doi.org/10.3389/fpls.2022.833030, 2022.
Mu, Z., Zeng, J., Zhang, Y., Song, W., Pang, W., Yi, Z., Asensio, D., Llusia, J., Penuelas, J., and Wang, X.: Soil uptake of isoprenoids in a Eucalyptus urophylla plantation forest in subtropical China, Front. For. Glob. Change, 6, 1260327, https://doi.org/10.3389/ffgc.2023.1260327, 2023.
Nagalingam, S., Seco, R., Kim, S., and Guenther, A.: Heat stress strongly induces monoterpene emissions in some plants with specialized terpenoid storage structures, Agric. For. Meteorol., 333, 109400, https://doi.org/10.1016/j.agrformet.2023.109400, 2023.
Niinemets, U., Kuhn, U., Harley, P. C., Staudt, M., Arneth, A., Cescatti, A., Ciccioli, P., Copolovici, L., Geron, C., Guenther, A., Kesselmeier, J., Lerdau, M. T., Monson, R. K., and Penuelas, J.: Estimations of isoprenoid emission capacity from enclosure studies: measurements, data processing, quality and standardized measurement protocols, Biogeosciences, 8, 2209–2246, https://doi.org/10.5194/bg-8-2209-2011, 2011.
Nunes, T. V. and Pio, C. A.: Emission of volatile organic compounds from Portuguese Eucalyptus forests, Chemosphere, 3, 239–248, https://doi.org/10.1016/S1465-9972(01)00007-1, 2001.
Peñuelas, J. and Staudt, M.: BVOCs and global change, Trends Plant Sci., 15, 133–144, https://doi.org/10.1016/j.tplants.2009.12.005, 2010.
Pfannerstill, E. Y., Arata, C., Zhu, Q., Schulze, B. C., Ward, R., Woods, R., Harkins, C., Schwantes, R. H., Seinfeld, J. H., Bucholtz, A., Cohen, R. C., and Goldstein, A. H.: Temperature-dependent emissions dominate aerosol and ozone formation in Los Angeles, Science, 384, 1324–1329, https://doi.org/10.1126/science.adg8204, 2024.
Pollastri, S., Jorba, I., Hawkins, T. J., Llusia, J., Michelozzi, M., Navajas, D., Penuelas, J., Hussey, P. J., Knight, M. R., and Loreto, F.: Leaves of isoprene-emitting tobacco plants maintain PSII stability at high temperatures, New Phytol., 223, 1307–1318, https://doi.org/10.1111/nph.15847, 2019.
Pollastri, S., Baccelli, I., and Loreto, F.: Isoprene: An antioxidant itself or a molecule with multiple regulatory functions in plants?, Antioxidants, 10, 684, https://doi.org/10.3390/antiox10050684, 2021.
Purser, G., Heal, M. R., White, S., Morison, J. I. L., and Drewer, J.: Differences in isoprene and monoterpene emissions from cold-tolerant eucalypt species grown in the UK, Atmos. Poll. Res., 11, 2011–2021, https://doi.org/10.1016/j.apr.2020.07.022, 2020.
Purser, G., Drewer, J., Morison, J. I. L., and Heal, M. R.: A first assessment of the sources of isoprene and monoterpene emissions from a short-rotation coppice Eucalyptus gunnii bioenergy plantation in the UK, Atmos. Environ., 262, 118617, https://doi.org/10.1016/j.atmosenv.2021.118617, 2021.
Satake, A., Hagiwara, T., Nagano, A. J., Yamaguchi, N., Sekimoto, K., Shiojiri, K., and Sudo, K.: Plant molecular phenology and climate feedbacks mediated by BVOCs, Annu. Rev. Plant Biol., 75, 605–627, https://doi.org/10.1146/annurev-arplant-060223-032108, 2024.
Scott, C. E., Arnold, S. R., Monks, S. A., Asmi, A., Paasonen, P., and Spracklen, D. V.: Substantial large-scale feedbacks between natural aerosols and climate, Nat. Geosci., 11, 44–48, https://doi.org/10.1038/s41561-017-0020-5, 2017.
Sharkey, T. D. and Singsaas, E. L.: Why plants emit isoprene, Nature, 374, 769–769, https://doi.org/10.1038/374769a0, 1995.
Sørensen, M., Rinnan, R., Woodrow, I., Møller, B. L., and Neilson, E. H. J.: The entangled dynamics of eucalypt leaf and flower volatile emissions, Environ. Exp. Bot., 176, 104032, https://doi.org/10.1016/j.envexpbot.2020.104032, 2020.
Staudt, M. and Visnadi, I.: High chemodiversity in the structural and enantiomeric composition of volatiles emitted by Kermes oak populations in Southern France, Elem. Sci. Anth., 11, 1, https://doi.org/10.1525/elementa.2023.00043, 2023.
Street, R. A., Hewitt, C. N., and Mennicken, S.: Isoprene and monoterpene emissions from a Eucalyptus plantation in Portugal, J. Geophys. Res.-Atmos., 102, 15875–15887, https://doi.org/10.1029/97jd00010, 1997.
Tsui, J. K.-Y., Guenther, A., Yip, W.-K., and Chen, F.: A biogenic volatile organic compound emission inventory for Hong Kong, Atmos. Environ., 43, 6442–6448, https://doi.org/10.1016/j.atmosenv.2008.01.027, 2009.
Wang, P., Zhang, Y., Gong, H., Zhang, H., Guenther, A., Zeng, J., Wang, T., and Wang, X.: Updating biogenic volatile organic compound (BVOC) emissions with locally measured emission factors in South China and the effect on modeled ozone and secondary organic aerosol production, J. Geophys. Res.-Atmos., 128, e2023JD039928, https://doi.org/10.1029/2023jd039928, 2023.
Weber, J., King, J. A., Abraham, N. L., Grosvenor, D. P., Smith, C. J., Shin, Y. M., Lawrence, P., Roe, S., Beerling, D. J., and Martin, M. V.: Chemistry-albedo feedbacks offset up to a third of forestation's CO2 removal benefits, Science, 383, 860–864, https://doi.org/10.1126/science.adg6196, 2024.
Wiedinmyer, C., Guenther, A., Harley, P., Hewitt, C.N., Geron, C., Artaxo, P., Steinbrecher, R., and Rasmussen, R.: Global organic emissions from vegetation, in: Emissions of Atmospheric Trace Compounds, edited by: Granier, C., Artaxo, P., and Reeves, C. E., Kluwer Publishing Co, Dordrecht, The Netherlands, 115–170, https://doi.org/10.1007/978-1-4020-2167-1_4, 2004.
Winters, A. J., Adams, M. A., Bleby, T. M., Rennenberg, H., Steigner, D., Steinbrecher, R., and Kreuzwieser, J.: Emissions of isoprene, monoterpene and short-chained carbonyl compounds from Eucalyptus spp. in southern Australia, Atmos. Environ., 43, 3035–3043, https://doi.org/10.1016/j.atmosenv.2009.03.026, 2009.
Yuan, X., Feng, Z., Shang, B., Calatayud, V., and Paoletti, E.: Ozone exposure, nitrogen addition and moderate drought dynamically interact to affect isoprene emission in poplar, Sci. Total Environ., 734, https://doi.org/10.1016/j.scitotenv.2020.139368, 2020
Zeng, J.: Measurement report: Age-dependent BVOC emissions in Eucalyptus urophylla: a comparison of leaf cuvette and branch chamber measurements, Mendeley [data set], https://doi.org/10.17632/jw8g8gkm5t.1, 2025
Zeng, J., Zhang, Y., Pang, W., Ran, H., Guo, H., Song, W., and Wang, X.: Optimizing in-situ measurement of representative BVOC emission factors considering intraspecific variability, Geophys. Res. Lett., 51, e2024GL108870, https://doi.org/10.1029/2024gl108870, 2024.
Zeng, J., Zhang, Y., Pang, W., Ran, H., Guo, H., Lu, Y., Song, W., and Wang, X.: Heatwaves suppress isoprene emission optima in subtropical Eucalyptus: Implications for biogenic VOC modeling under extreme thermal events, Geophys. Res. Lett., 52, e2025GL114767, https://doi.org/10.1029/2025GL114767, 2025a.
Zeng, J., Zhang, Y., Pang, W., Ran, H., Mu, Z., Guo, H., Lu, Y., Song, W., and Wang, X.: Decoupling temperature and light effects on terpene emissions from subtropical Eucalyptus: Insights from controlled field measurements, J. Geophys. Res.-Atmos., 130, e2024JD042616, https://doi.org/10.1029/2024JD042616, 2025b.
Zeng, J., Zhang, Y., Ran, H., Pang, W., Guo, H., Mu, Z., Song, W., and Wang, X.: Calibrating adsorptive and reactive losses of monoterpenes and sesquiterpenes in dynamic chambers using deuterated surrogates, Atmos. Meas. Tech., 18, 1811–1821, https://doi.org/10.5194/amt-18-1811-2025, 2025c.
Zeng, J., Zhang, Y., Pang, W., Ran, H., Mu, Z., Guo, H., Lu, Y., Song, W., and Wang, X.: Contrasting Emission seasonality between light-dependent and light-independent biogenic VOCs from subtropical Eucalyptus trees, J. Geophys. Res.-Atmos., 130, e2025JD043387, https://doi.org/10.1029/2025jd043387, 2025d.
Zeng, J., Song, W., Zhang, Y., Mu, Z., Pang, W., Zhang, H., and Wang, X.: Emissions of isoprenoids from dominant tree species in subtropical China, Front. For. Glob. Change, 5, 1089676, https://doi.org/10.3389/ffgc.2022.1089676, 2022b.
Zeng, J., Zhang, Y., Zhang, H., Song, W., Wu, Z., and Wang, X.: Design and characterization of a semi-open dynamic chamber for measuring biogenic volatile organic compound (BVOC) emissions from plants, Atmos. Meas. Tech., 15, 79–93, https://doi.org/10.5194/amt-15-79-2022, 2022a.
Zeng, J., Zhang, Y., Mu, Z., Pang, W., Zhang, H., Wu, Z., Song, W., and Wang, X.: Temperature and light dependency of isoprene and monoterpene emissions from tropical and subtropical trees: Field observations in south China, Appl. Geochem., 155, 105727, https://doi.org/10.1016/j.apgeochem.2023.105727, 2023.
Zhang, Y., Zhang, R., Yu, J., Zhang, Z., Yang, W., Zhang, H., Lyu, S., Wang, Y., Dai, W., Wang, Y., and Wang, X.: Isoprene mixing ratios measured at twenty sites in China during 2012-2014: Comparison with model simulation, J. Geophys. Res.-Atmos., 125, e2020JD033523, https://doi.org/10.1029/2020jd033523, 2020.
Zuo, Z., Weraduwage, S. M., Lantz, A. T., Sanchez, L. M., Weise, S. E., Wang, J., Childs, K. L., and Sharkey, T. D.: Isoprene acts as a signaling molecule in gene networks important for stress responses and plant growth, Plant Physiol., 180, 124–152, https://doi.org/10.1104/pp.18.01391, 2019.