the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Widespread occurrence of large molecular methylsiloxanes in ambient aerosols
Beatriz Sayuri Oyama
Agne Masalaite
Dipayan Paul
Haiyan Ni
Hanne Noto
Dušan Materić
Maria de Fátima Andrade
Ru-Jin Huang
Ulrike Dusek
Synthetic pollutants have emerged as a widespread environmental concern. Recently, large molecular methylsiloxanes were identified in traffic emissions. Here, we show that large molecular methylsiloxanes are widely present in atmospheric particulate matter across diverse environments, including urban, coastal, rural, and forest sites in the Netherlands, Lithuania, and Brazil. Overall, methylsiloxanes of varying molecular sizes account for approximately 2.0 %–4.3 % of the non-refractory organic aerosol mass detected by thermal desorption proton transfer reaction mass spectrometry (TD-PTR-MS) analysis. Thermal desorption profiles indicate that over half of the detected methylsiloxanes originate from the depolymerization of large molecular methylsiloxanes, primarily associated with traffic emissions, while the remainder likely arise from the gas-to-particle conversion of volatile methylsiloxanes. Large molecular methylsiloxanes show a distinct correlation with long-chain hydrocarbons characteristic of engine lubricants, suggesting a lubricant-related source. Notably, the mass fraction of methylsiloxanes in organic aerosols does not decrease significantly during atmospheric transport and dilution, and a substantial fraction persists as large molecular methylsiloxanes. This persistence underscores their chemical stability, in contrast to the co-emitted lubricant hydrocarbons that undergo atmospheric oxidation. The substantial mass fraction of methylsiloxanes in particulate matter highlights their role as one of the most concentrated categories of synthetic compounds in the atmosphere, raising concerns about their potential, yet poorly understood, effects on human health and the climate.
- Article
(2326 KB) - Full-text XML
-
Supplement
(4463 KB) - BibTeX
- EndNote
Synthetic materials have improved human convenience, yet their pervasive presence in the environment raises concerns for ecosystems and human health. Recent attention has focused on certain synthetics, notably microplastics (Jambeck et al., 2015) and per- and polyfluoroalkyl substances (PFAS) (Evich et al., 2022), with global annual productions of 390.7 (PlasticsEurope, 2022) and 0.23 million t (Glüge et al., 2020), respectively. Siloxanes, another class of synthetic compounds extensively used in/as lubricants, had a global annual production of approximately 6.59 million metric tons in 2025 (Claight Corporation, 2026). These synthetic organic compounds feature alternating silicon and oxygen arrangements in their skeletons, predominantly manifesting as methylsiloxanes (Rücker and Kümmerer, 2015). Despite the fact that some volatile species have been proved to be health hazards (US EPA, 2022; ECHA, 2024), such as affecting estrogen and liver (Quinn et al., 2006; Franzen et al., 2017; Gentry et al., 2017; Franzen et al., 2016), methylsiloxanes have received limited attention. Polydimethylsiloxanes (PDMS, or silicone oil; CH3[Si(CH3)2O]nSi(CH3)3) are commonly used in small amounts as defoaming agents in lubricants and can sometimes function directly as lubricants. Cyclic volatile methylsiloxanes (cVMS; ((Si(CH3)2O)n, ), with their “roll-like-a-wheel” structure, can commonly serve as lubricants themselves. Given their extensive use, methylsiloxanes released into the atmosphere may have implications for both human health and climate, yet these potential impacts remain poorly studied.
Until recently, the main origins of methylsiloxanes detected in the atmosphere were attributed to the volatilization of small molecular methylsiloxanes in personal care and industrial products (Tang et al., 2015; Yucuis et al., 2013; Coggon et al., 2018). Consequently, prior research predominantly focused on gas-phase volatile methylsiloxanes (VMS), while those in the particle phase were mainly considered to be byproducts of gas-to-particle transfer (Bzdek et al., 2014; Janechek et al., 2019; Han et al., 2022). However, our recent studies have revealed substantial quantities of particle-phase methylsiloxanes emitted from ships (Yao et al., 2022) and vehicles (Yao et al., 2023), indicating far greater emissions of methylsiloxanes than previously presumed. Notably, the methylsiloxanes emitted by ships and vehicles include a significant fraction of large molecular methylsiloxanes, which are not directly detectable by conventional mass spectrometry due to their high molecular weights but can be identified following high-temperature thermal depolymerization into smaller fragments. This differs from previously identified small molecular VMS added in personal care and industrial products, representing a novel pollutant that has remained undetected. Therefore, the overall presence of methylsiloxanes in the atmospheric aerosols may have been significantly underestimated, resulting human inhalation and fate of these small and large molecular methylsiloxanes being poorly understood.
Employing a high-resolution mass spectrometry analysis technique based on the characteristic peak distribution of natural silicon isotopes (Yao et al., 2022, 2023), our objective is to quantify methylsiloxanes in atmospheric particulate matter across diverse environments and identify their primary source. Particular attention is given to large molecular methylsiloxanes and their co-emission with long-chain hydrocarbons from lubricants, which may provide insights into atmospheric processes. We further explore the human inhalation and potential environmental impacts of large molecular methylsiloxanes.
2.1 Sampling of atmospheric particulate matter
Particulate matter samples were collected from diverse environments. In the Netherlands, sampling was carried out at the Cesar Observatory (51.97° N, 4.90° E) (Dusek et al., 2017), a rural site situated in a heavily industrialized region in Western Europe, located between The Hague, Rotterdam, Amsterdam, and Utrecht. Particulate matter samples with sizes smaller than 2.5 µm (PM2.5) were collected on pre-cleaned quartz filters (Whatman, QM-A; heated at 800 °C for 12 h) using a high-volume sampler (Digitel DHA-80) and a flow rate of 500 L min−1. Samples were collected separately for day and night over a period of several days during different seasons (n=38), specifically from 11 February 2011 to 16 October 2012, and the sampling duration varied by season, ranging from 2 to 9 d. Before and after sampling, the filters were kept wrapped in pre-baked aluminum foil and stored at −20 °C.
Aerosol samples were collected at three representative sites in Lithuania, i.e., Vilnius (urban, 54.63° N, 25.17° E), Preila (coastal, 55.37° N, 21.02° E), and Rugsteliskis (forest, 55.45° N, 26.00° E), representing typical northern European environments. Sampling campaigns were carried out during winter, when ambient temperatures averaged around −4 °C: from 30 December 2008 to 26 January 2009 in Vilnius, 8–15 December 2012 in Preila, and 1–29 March 2013 simultaneously at all three sites.
During the 2008–2009 and 2012 campaigns (Masalaite et al., 2018), PM1 aerosol samples were collected at Vilnius (n=5) and Preila (n=4) using a micro-orifice uniform deposition impactor (MOUDI-110) equipped with 11 stages and operated at a flow rate of 30 L min−1. Aluminum foils (47 mm diameter) were used as collection substrates and preheated at 600 °C for 10 h to remove trace organic contaminants prior to sampling. Although this temperature was relatively high for aluminum, no abnormal effects were observed in final results.
In March 2013, PM1 aerosol samples were collected simultaneously at all three sites (Masalaite et al., 2017). At Vilnius (n=19), samples were collected on 150 mm quartz microfiber filters (Whatman QM-A) using a high-volume sampler (Digitel DH-77) operating at 500 L min−1. At Preila (n=6), a low-volume sampler (Leckel) operating at 30 L min−1 was used with 47 mm quartz filters, while at Rugsteliskis (n=6), samples were collected on 75 mm aluminum foils using a high-flow, three-stage impactor (MOUDI 128) operated at 100 L min−1. Filters and foils were preheated at 550 °C for 12 h prior to sampling to eliminate residual organics. After collection, the samples were wrapped in pre-fired aluminum foil (500 °C, 12 h), sealed in plastic bags, and stored at −25 °C until analysis.
In Brazil, particulate matter samples were collected in São Paulo (23.56° S, 46.73° W) (Oyama et al., 2016), a large metropolitan area in South America. Daily ambient samples were collected during the winter of Southern Hemisphere, specifically from 6 July to 9 September 2012, with an average temperature of approximately 18 °C. PM2.5 samples (n=31) were collected on pre-cleaned quartz filters (Whatman, QM-A; heated at 800 °C for 12 h) using a high-volume sampler and a flow rate of 1.13 m3 min−1. Following the sampling procedure, the collected samples were immediately enveloped in pre-cleaned aluminum foil (550 °C for 8 h), sealed within polyethylene bags, and subsequently stored in low-temperature freezers at −18 °C until analysis.
Despite differences in sampling methods and particle size cutoffs, we do not expect these factors to substantially affect the derived methylsiloxane fractions. It should be noted that PM1 typically accounts for 60 %–90 % of the PM2.5 mass (Theodosi et al., 2011; Liu et al., 2024); therefore, the methylsiloxane concentrations in Lithuanian PM1 aerosols are expected to be approximately 10 %–40 % lower than those in PM2.5 samples from other regions.
2.2 Chemical analysis
The chemical composition analysis of the collected particulate matter samples was conducted using a thermal desorption – proton transfer reaction – time of flight – mass spectrometer (TD-PTR-ToF-MS, PTR-TOF8000, Ionicon Analytik GmbH, Austria) (Holzinger et al., 2010a, b) with a mass resolution of 3000–4000 at full width at half maximum. The particulate matter samples were thermally desorbed from the filters using an oven with temperature steps of 3 min from 100 to 350 °C in 50 °C increments for the Lithuanian and Brazilian samples, and of 3 min from 125 to 350 °C in 75 °C increments for the samples collected in the Netherlands. A pure nitrogen carrier gas at a flow rate of 100 or 50 mL min−1 was used for carrying the desorption products to the PTR-MS. To prevent condensation of organic compounds, the drift tube and inlet line temperatures were maintained at 120 and 180 °C, respectively. A higher temperature could further reduce the risk of condensation losses, but increasing the drift tube temperature further is currently technically challenging for us.
Based on prior thermal tests of PDMS with different molecular weights (Yao et al., 2023), PDMS with a viscosity of 10 cSt, dominated by molecules containing approximately 15 siloxane units (with characteristic ions at for cyclic species), exhibits its main desorption/decomposition peak slightly below 200 °C. In contrast, higher-molecular-weight PDMS (e.g., 20 cSt, ∼25 siloxane units) shows its dominant peak above 200 °C. Moreover, methylsiloxanes containing ∼15 siloxane units have extremely low volatility under ambient conditions. Therefore, we suggest ∼15 siloxane units and 200 °C as practical thresholds for defining large molecular methylsiloxanes.
In addition, we focused on the desorbed organic aerosol fraction up to 350 °C, hereafter referred to as non-refractory organic aerosols (OA). We did not investigate the non-desorbed fraction due to the technical limitation of our custom-designed heating unit. Previous work has shown that large molecular methylsiloxanes, such as PDMS with 10 000 cSt and 800 siloxane units, can withstand temperatures exceeding 650 °C (Yao et al., 2023). Consequently, part of the large molecular methylsiloxanes may not have undergone complete thermal decomposition in our experimental conditions, and both methylsiloxane and total organic aerosol concentrations reported here should be considered lower-bound estimates. Detailed discussion regarding thermal desorption and PTR-MS analysis of organics refers to our previous studies (Holzinger et al., 2010b; Timkovsky et al., 2015; Oyama et al., 2016; Materić et al., 2017).
PTRwid software was employed to identify and integrate peaks (Holzinger, 2015) in the mass spectra, to give the concentration of individual compounds in ppb in the carrier gas. The instrument was initially calibrated with standards to establish a transmission curve (Fig. S9), which was then applied to convert signal intensity into concentration (Holzinger et al., 2019; Worton et al., 2023). A unified mass list comprised the mass-to-charge ratio () with ±2 SD (standard deviations) (95 % confidence interval) for all identified peaks, along with potential molecular formulas having within this range of uncertainty. The median values of the system blanks and the field blanks were subtracted from the mass spectra. Most samples were analyzed in triplicate, with median concentration values used for further calculations. Detection limits were typically calculated as three times the standard deviation of the blank and account for ∼1.5 % of sample concentrations on average. The uncertainty is approximately 20.3 % of the measured concentration, evaluated as the ratio of the standard deviation to the mean of replicate measurements. Due to the relatively large variability in sample concentrations, we assess uncertainty using this percentage-based approach. This level of uncertainty is slightly higher than that of conventional mass spectrometry methods but is typical for TD-PTR-MS (Holzinger et al., 2010b), as thermal desorption processes and protonation probabilities can introduce additional variability.
2.3 Identification and quantification of methylsiloxanes
Upon heating of the filter samples, smaller methylsiloxanes contained in the aerosol particles thermally desorb at relatively low temperatures. On the other hand, large molecular methylsiloxanes (e.g., PDMS) undergo depolymerization into small volatile methylsiloxanes (VMS) and fragments, with characteristic concentration ratios, dominated by D3, followed by D4, D5, and others (Yao et al., 2023; Thomas and Kendrick, 1969; Camino et al., 2001). The VMS resulting from both desorption and depolymerization were detected by the PTR-MS and identified based on the abundance of silicon isotopes, i.e., 28Si ( amu, 92.223 %), 29Si ( amu, 4.685 %), and 30Si ( amu, 3.092 %) (Yao et al., 2022, 2023). The isotope peaks of methylsiloxane molecules exhibit significantly higher intensities compared to other organic compounds with CHON structures, owing to the high abundance of 29Si and 30Si isotopes. For example, the main peak of D5 (C10H30O5Si5) is observed at . The first isotope peak ranges from to 372.106 and accounts for 36.41 % of the main peak height, and the second isotope peak ranges from to 373.108 and accounts for 23.56 % of the main peak height. In contrast, organic molecules with CHON structures exhibit lower isotope peaks. For instance, the main peak of C11H18O12N2 at cannot be distinguished from D5 with the mass resolution of PTR-MS. However, the first isotope peak of C11H18O12N2 only reaches 13.29 % of the main peak height, and the second isotope peak only reaches 3.25 %. With the characteristic ratios of the first and second isotope peaks to the main peak, various methylsiloxane depolymerization products and their derivatives were identified, including cVMS (D3–D10) and positively charged fragments, monomer fragments (CH3)2(OH)2Si and (CH3)2(OH)Si+ (DMSD and DMHS+), and hydroxylated methylsiloxanes and related positively charged fragments. The quantification approach is based primarily on minimizing interference of other chemical compounds occurring at either the same as the main peak or as the isotope peaks (Yao et al., 2022, 2023). Details on the identification and quantification of methylsiloxanes are given in Method S1. The identified methylsiloxane molecules resulting from desorption and depolymerization of the large molecular methylsiloxanes in each aerosol sample are shown in Figs. S1–S6 and Sects. S1–S4 in the Supplement.
2.4 Estimation of human inhalation rate
The inhalation rate is determined by multiplying the inhalation volume by the pollutant concentration per unit volume. The typical 24 h inhalation volumes for children and adults were sourced from previous studies (Allan and Richardson, 1998; Stifelman, 2007). Methylsiloxane concentrations in particulate matter were derived from the results of this study. Concentrations of PFAS were taken from established literature sources (Faust, 2023). See Method S2 for details.
Atmospheric microplastics are conventionally reported within a size range of 1–5000 µm in diameter, focusing on particle number concentrations. To evaluate mass concentrations of inhalable microplastics (1–10 µm), a custom model was developed based on a combination of assumptions and previously reported data related to microplastic particle number concentrations, morphological characteristics, size distribution, and density distribution. There is currently no consensus on whether nanoplastic concentrations in the sub-1 µm size range are higher or lower than those of microplastics. Consequently, mass concentrations of nanoplastics up to 1 µm in size were estimated by presuming that they exhibit similar PM1 to PM10 ratios as found in particulate matter. More details are available in Model S1.
3.1 Methylsiloxanes in atmospheric particulate matter
Although methylsiloxanes in atmospheric aerosols have been studied to some extent, knowledge about large molecular methylsiloxanes, compounds that only undergo thermal depolymerization at high temperatures, remains almost entirely lacking. To address this gap, we quantified particle-phase methylsiloxanes across a range of diverse environments, including coastal, urban, rural, and forest regions. Sampling locations were conducted in the Netherlands (West Europe), Lithuania (Northeastern Europe), and São Paulo in Brazil (South America). Concentrations of particle-phase methylsiloxanes and their mass fractions within non-refractory organic aerosols (OA) were determined in these diverse environments (Fig. 1a and b).
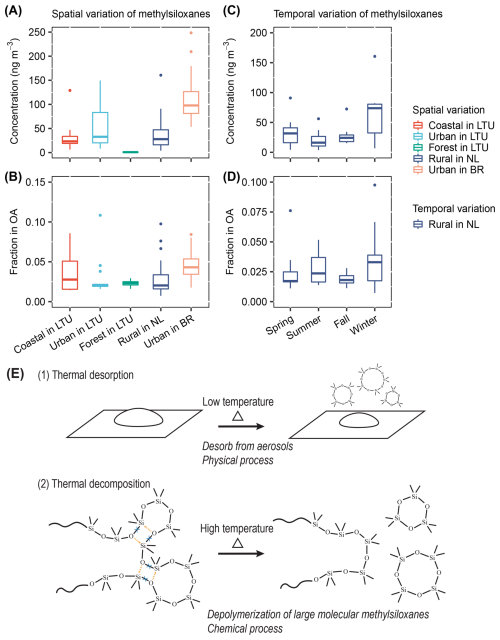
Figure 1Spatial and temporal variations of methylsiloxanes in atmospheric particulate matter (PM). Concentrations (a) and fractions (b) of methylsiloxanes in non-refractory organic aerosols (OA, thermally-desorbed up to 350 °C) at various locations: coastal (number of samples n=11, winter), urban (n=23, winter), forest (n=6, winter) in Lithuania (LTU; PM1); rural in the Netherlands (NL; n=38, four seasons; PM2.5); and urban in São Paulo, Brazil (BR; n=31, winter; PM2.5). Concentrations (c) and fractions (d) of methylsiloxanes in organic aerosols during different seasons in the Netherlands. (e) Processes occurring during sample heating for TD-PTR-MS analysis: (1) thermal desorption of small molecular methylsiloxanes and (2) thermal depolymerization of large molecular methylsiloxanes contained in the aerosol samples.
Methylsiloxanes are widespread across diverse environments and occur at substantial concentrations (Fig. 1). It should be noted that the Lithuanian samples represent PM1, whereas the samples from the Netherlands and Brazil correspond to PM2.5. Since PM1 typically accounts for 60 %–90 % of PM2.5 mass (Theodosi et al., 2011; Liu et al., 2024), the methylsiloxane concentrations in Lithuanian aerosols are expected to be 10 %–40 % lower. The median concentrations of methylsiloxanes were highest in urban areas, 33 ng m−3 in Lithuania (PM1) and 98 ng m−3 in Brazil (PM2.5), and lowest in the forest site (0.9 ng m−3 in Lithuania, PM1). Intermediate levels were observed in coastal (23 ng m−3 in Lithuania, PM1) and rural environments (28 ng m−3 in the Netherlands, PM2.5). Generally, locations with higher population densities exhibited higher concentrations of methylsiloxanes, whereas locations with lower population densities showed lower concentrations. This observation can be explained by higher traffic emissions in the urban environment or/and higher residential indoor emissions. The median mass fractions of methylsiloxanes in OA varied from 2.0 % in rural areas of the Netherlands to 4.3 % in urban areas of Brazil. These values surpass those observed in primary emissions from ships (1.2 %) (Yao et al., 2022) and vehicles (1.1 %) (Yao et al., 2023). This enrichment suggests either higher atmospheric stability of the methylsiloxanes than other primary aerosol matter and/or additional particulate methylsiloxane formation via additional sources or secondary processes.
Filter samples covering a whole seasonal cycle were analyzed in the Netherlands, allowing us to determine the seasonal variations of methylsiloxanes in atmospheric particulate matter (Fig. 1c and d). The median concentrations of methylsiloxanes were lowest in summer (16 ng m−3) and highest in winter (74 ng m−3) in the rural Netherlands, indicating significant differences (1-way ANOVA, p=0.01). The median fractions of methylsiloxanes in OA varied between 1.7 % (spring) and 3.3 % (winter), remaining relatively stable throughout the year. These results suggest that, although particle-phase methylsiloxanes exhibit seasonal variability, they represent a continuous and persistent environmental concern throughout the year.
The heating of filter samples for PTR-MS analysis (Fig. 1e) involves two main processes: (i) thermal desorption of small molecular methylsiloxanes and their oxidized products, and (ii) thermal depolymerization of large molecular methylsiloxanes. Accordingly, the total particle-phase methylsiloxanes include contributions from both components, which will be further discussed later. VMS are primarily distributed in the gas phase due to their relatively high vapor pressures, partitioning mainly to the gas phase rather than the particle phase. Oxidation lowers vapor pressures of VMS and promotes gas-to-particle partitioning; however, even oxidized VMS remain small molecules that volatilize upon mild heating. During thermal desorption, these small molecular methylsiloxanes and the associated oxidized products evaporate at relatively low temperatures from the aerosol filter sample. At high temperatures (e.g., >200 °C), large molecular methylsiloxanes (e.g., PDMS) undergo depolymerization, yielding smaller cVMS (D3–Dn) (Yao et al., 2023; Thomas and Kendrick, 1969; Camino et al., 2001) and other by-products. This thermal depolymerization was evident in our data, as small molecular cVMS are too volatile to persist in the particle phase at such high temperatures. Thus, the high-temperature signal reflects large molecular methylsiloxanes, which differ fundamentally from gas-to-particle VMS both in properties and in sources.
3.2 Atmospheric methylsiloxanes and traffic emissions
After initially detecting large molecular methylsiloxanes in traffic-related source samples (Yao et al., 2023), we now confirm their widespread and substantial presence in atmospheric aerosols from three countries and four diverse environments. Methylsiloxanes detected at high desorption temperatures (e.g., >200 °C) can be attributed almost entirely to large molecular methylsiloxanes, with traffic currently recognized as their only known source (additional sources cannot be excluded). In contrast, the low-temperature fractions contain both smaller fractions of large molecular methylsiloxanes (depolymerized ≤ 200 °C) and a considerable contribution from VMS that likely entered the particle phase through gas-to-particle conversion. These VMS can originate from household and industrial emissions and partition into aerosols via oxidation, adsorption, and related processes.
To preliminarily distinguish these sources in aerosols, we compared the concentrations and relative mass fractions of methylsiloxanes detected at different desorption temperature steps in traffic sources (vehicle and ship) and ambient aerosol samples (Fig. 2). As outlined in the thermal desorption and depolymerization mechanism (Fig. 1e), the species observed across all temperature steps were mainly smaller cVMS (D3–Dn, Fig. 2a and b) and their characteristic fragments resulting from the loss of a −CH3 group (Yao et al., 2022, 2023; Worton et al., 2023). We also identified dimethylsilanediol (DMSD, (CH3)2(OH)2Si) and its associated fragment dimethylhydroxysilyl cation (DMHS+, (CH3)2(OH)Si+), consistent with monomeric diols produced via hydrolysis of methylsiloxanes. Small amounts of hydroxylated methylsiloxanes were detected as well, and their concentrations were grouped with the major cVMS (see Method S1 for details).
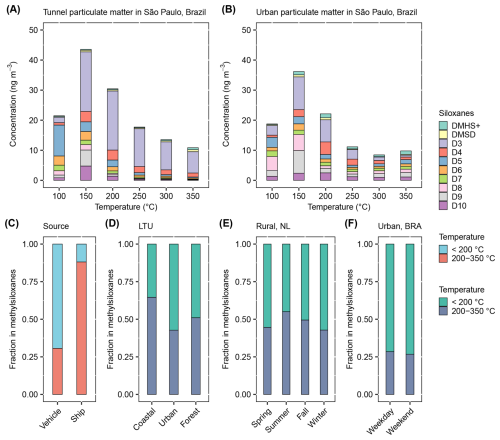
Figure 2Characteristics of methylsiloxanes detected from heating traffic and atmospheric particulate matter samples at various desorption temperatures. Detected methylsiloxanes include desorbed molecules as well as depolymerization products of large molecular methylsiloxanes. Concentrations of various methylsiloxanes as a function of desorption temperature in (a) tunnel and (b) urban particulate matter in São Paulo, Brazil. The relative mass fraction of methylsiloxanes released at temperatures ≤ 200 and 200–350 °C for (c) particulate matter sampled in two tunnels (vehicle) and from the smoke stack of a high-speed passenger hydrofoil (ship); (d) particulate matter sampled at coastal, urban, and forest sites in Lithuania; (e) particulate matter sampled at a rural site in the Netherlands across various seasons; and (f) particulate matter sampled in urban São Paulo, Brazil.
A direct comparison of source and ambient aerosols from the same region (São Paulo, Brazil) illustrates how traffic emitted large molecular methylsiloxanes mix with gas-to-particle VMS in the atmosphere. In tunnel samples, methylsiloxane concentrations varied systematically with desorption temperature, and similar trends were evident in ambient aerosols (Fig. 2a and b, fractions in Fig. S6). Previous studies have shown that gaseous VMS typically follow the distribution D5 > D4 > D3 (Yucuis et al., 2013; Genualdi et al., 2011; Brunet et al., 2024). However, PDMS depolymerization usually yields predominantly D3, followed by D4, D5, and others (D3 > D4 > D5; Yao et al., 2023; Thomas and Kendrick, 1969; Camino et al., 2001). These contrasting trends were evident across different temperature steps during thermal analysis. In both tunnel and ambient aerosols, D5 was the most abundant methylsiloxane at a desorption temperature of 100 °C, consistent with gas-to-particle conversion. Above 150 °C, D3 becomes the most abundant methylsiloxane, highlighting contributions from depolymerization of large molecular methylsiloxanes. At 150 °C, the concentrations of D8–D10 increased compared to 100 °C, suggesting an additional contribution from gas-to-particle conversion, where larger molecules (D8–D10) desorb at slightly higher temperatures than smaller molecules (D5–D7). In contrast, methylsiloxane fractions desorbed above 200 °C were dominated almost exclusively by large molecular methylsiloxanes, confirming that these high-temperature signals represent a new class of pollutants. Overall, tunnel source samples were characterized primarily by large molecular methylsiloxanes with minor contributions from gas-to-particle conversion. In contrast, ambient aerosols in the same region contained strong signatures from both sources, with large molecular methylsiloxanes contributing slightly more than VMS originating from gas-to-particle conversion.
Because thermal desorption signatures of gas-to-particle VMS have not yet been systematically characterized and no reliable source profiles exist, a full source apportionment of aerosol-phase methylsiloxanes is not currently possible. Nevertheless, since ≤200 °C fractions are contributed by both gas-to-particle VMS and large molecular methylsiloxanes, whereas 200–350 °C fractions are composed almost exclusively of large molecular methylsiloxanes, this temperature threshold provides a practical way to broadly assess their relative contributions across different regions. The relative mass fractions of methylsiloxanes detected at lower (≤200 °C, both gas-to-particle VMS and large molecular methylsiloxanes) versus higher temperatures (200–350 °C, predominantly large molecular methylsiloxanes) were compared for samples from traffic sources and ambient atmospheric aerosols (Fig. 2c–f). The depolymerization of large molecular methylsiloxanes is temperature-dependent, with larger molecules requiring higher temperatures to break down. Methylsiloxanes from ship emissions (Fig. 2c) exhibited depolymerization predominantly at 200–350 °C, whereas methylsiloxanes from vehicle emissions predominantly at ≤200 °C. These results imply that ships emit a higher proportion of larger methylsiloxane molecules than vehicles, attributed to differences in engine design and lubrication (Yao et al., 2023).
In Lithuania, the mass fractions of methylsiloxanes at 200–350 °C were higher at a coastal site downwind of a harbor than in urban locations (Fig. 2d), consistent with a higher contribution of ship emissions at the coastal location and a higher contribution of vehicle emissions in the urban region. Dutch samples (Fig. 2e) were collected at a rural background station, located between major harbor cities (Rotterdam, Amsterdam, and The Hague) and inland cities (Utrecht and Eindhoven). At this site, mass fractions of methylsiloxanes at 200–350 °C were lower in winter compared to other seasons. Air mass back trajectory analysis (Fig. S11) revealed that winter air masses were primarily continental, whereas in other seasons they arrived from the west over the ocean. This seasonal contrast indicates that ship emissions substantially increase the high-temperature fraction outside of winter. In São Paulo, Brazil (Fig. 2f), the thermal desorption profiles of urban aerosols closely resembled those of vehicle source samples collected in two local tunnels (Fig. 2c). The strong similarity in desorption characteristics, with minor differences likely due to gas-to-particle transformation, indicates that vehicle emissions are the dominant source of methylsiloxanes in São Paulo's atmosphere.
Overall, large molecular methylsiloxanes represent a substantial fraction of atmospheric aerosols in both Lithuania and the Netherlands, where 200–350 °C fractions already accounted for more than half of the total. This highlights that these newly identified large molecular methylsiloxanes are far more abundant than previously recognized gas-to-particle VMS. In São Paulo, the 200–350 °C fraction contributed more than one-quarter of the total, and depolymerization signatures of large molecular methylsiloxanes were already apparent at 150 and 200 °C, together accounting for over half of the total.
3.3 Atmospheric transport, dilution, and oxidation
Our results demonstrate that large molecular methylsiloxanes are widely present in atmospheric aerosols across diverse environments. Since traffic emissions are the only currently known large-scale source, an important question is how these compounds disperse into remote regions such as rural or forested areas. During atmospheric transport, dilution inevitably occurs, while additional methylsiloxanes may enter the particle phase via gas-to-particle conversion of VMS, and oxidation processes may further alter their composition. However, the relative importance of these processes remains uncertain.
In our tunnel study (Yao et al., 2023), we identified large molecular methylsiloxanes and long-chain hydrocarbons (C23–C38) from lubricants as co-emitted components of traffic particulate matter. These long-chain hydrocarbons are distinct markers of traffic emissions because they differ from shorter-chain hydrocarbons in gasoline (C5–C12), diesel (C12–C20), or biogenic sources. Previous studies reported their origins from internal combustion engines in vehicles, ships, and aircraft (Eichler et al., 2017; Masalaite et al., 2018; Yao et al., 2023; Decker et al., 2024; Sonntag et al., 2012). The co-occurrence of large molecular methylsiloxanes and long-chain hydrocarbons provides a tracer framework to track the transport and transformation of traffic-derived large molecular methylsiloxanes.
A comparison of tunnel and ambient samples in São Paulo illustrates how these tracers evolve in the atmosphere (Fig. 3). While both methylsiloxanes and long-chain hydrocarbons were strongly correlated in tunnel samples (R2=0.83, Fig. S12), at the urban background site long-chain hydrocarbons decreased sharply, whereas methylsiloxanes declined much less. This divergence suggests faster atmospheric oxidation of long-chain hydrocarbons and greater persistence of large molecular methylsiloxanes, consistent with their higher chemical stability. Additional methylsiloxanes at the background site may also originate from gas-to-particle transformation (Bzdek et al., 2014; Janechek et al., 2019; Han et al., 2022). Although methylsiloxanes can undergo oxidation by OH and Cl radicals (Alton and Browne, 2020), our isotopic analysis captures their oxidation and hydrolysis products, indicating that most remain accounted for in the particle phase.
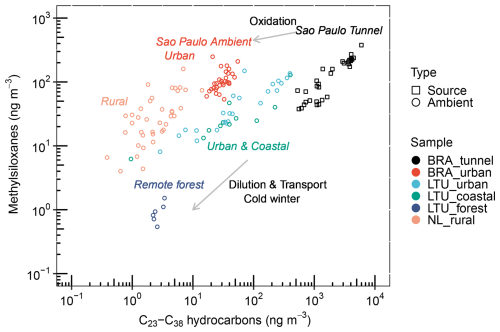
Figure 3Scatter plot of methylsiloxanes versus C23–C38 hydrocarbons in tunnel and atmospheric particulate matter. The tunnel particulate matter was collected in São Paulo (Brazil, BR), while ambient samples include atmospheric particulate matter collected in São Paulo, Lithuania (LTU), and the Netherlands (NL).
In Lithuania, atmospheric particulate matter was collected during a cold winter (average −4 °C). Despite the vastly different environments, methylsiloxanes and long-chain hydrocarbons showed similar correlations at urban, coastal, and forest sites, comparable to those observed in São Paulo tunnels. In contrast to São Paulo, the two compound classes decreased at comparable rates from polluted to clean sites, pointing to dilution as the dominant atmospheric process. The cold temperatures and weak solar radiation likely suppressed oxidation, allowing long-chain hydrocarbons to persist longer in ambient aerosols. At the rural background site in the Netherlands, methylsiloxanes levels were of the same order of magnitude (or slightly lower) than in Lithuania, while long-chain hydrocarbons reached their lowest concentrations across all sites, suggesting stronger oxidative loss under milder climatic conditions.
4.1 Methylsiloxanes inhalation
The potential health impacts of large molecular methylsiloxanes remain largely unknown and call for further investigation. Here, we provide a preliminary assessment of human exposure through inhalation, estimating the intake of aerosol-bound methylsiloxanes across different environments as a basis for future toxicological studies. Inhalation of atmospheric particulate matter, especially fine particles (<2.5 µm), can lead to deposition in the lungs. Although both short- and long-term health risks of such exposure remain uncertain, daily human intake can be estimated.
Our results show that inhalation rates of particle-phase methylsiloxanes vary substantially across environments (Fig. 4a). In Lithuania's forested areas, median daily intakes were 11.3 ng per capita per day for children and 13.0 ng per capita per day for adults, while in urban Brazil, values reached 1290 and 1480 ng per capita per day, respectively. Given the strong association of large molecular methylsiloxanes with traffic emissions, these results suggest that human exposure is a widespread and potentially significant issue on a global scale.
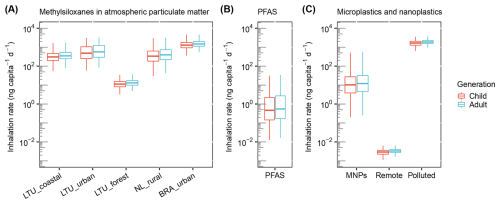
Figure 4Inhalation rates of methylsiloxanes in atmospheric particulate matter compared to other emerging pollutants. Estimated inhalation rates of (a) methylsiloxanes, (b) per- and polyfluoroalkyl substances (PFAS), and (c) micro- and nanoplastics (MNPs) in atmospheric particulate matter. Refer to Model S1 for details.
To contextualize our findings, we compared methylsiloxane inhalation with two other synthetic pollutants commonly reported in atmospheric particulate matter: PFAS and micro-/nanoplastics. Using literature-based aerosol concentrations of PFAS (Faust, 2023), we estimated median inhalation rates of 0.467 ng per capita per day for children and 0.550 ng per capita per day for adults (Fig. 4b), which are three to four orders of magnitude lower than the methylsiloxane intakes in urban and coastal regions reported in this study. For micro- and nanoplastics, we converted particle number concentrations into mass concentrations by accounting for particle size distributions, morphology, and density (details in Model S1). Monte Carlo simulations were then applied, combined with literature-derived ranges of particle abundance (Mohamed Nor et al., 2021; Revell et al., 2021), yielding median inhalation estimates of 10.3 ng per capita per day for children and 12.1 ng per capita per day for adults (Fig. 4c). These values are one to two orders of magnitude lower than the methylsiloxane levels in urban environments. These comparisons highlight that, while the toxicological properties of methylsiloxanes are far less understood than those of PFAS or micro-/nanoplastics, the levels of human intake through inhalation can be substantially higher. This underscores the importance of investigating their potential health risks and long-term environmental impacts.
4.2 Potential impacts of methylsiloxanes on aerosol properties
Methylsiloxanes are widely used in industrial formulations, notably as defoaming agents and anti-freeze components, where even small additions can alter the physicochemical properties of lubricants. In ambient aerosols from diverse environments, we detected substantial levels of large molecular methylsiloxanes, with total methylsiloxanes contributing 2.0 %–4.3 % of organic aerosol and the large molecular fraction comprising more than half. These findings indicate that large molecular methylsiloxanes may exert an appreciable influence on the physicochemical properties of atmospheric aerosols.
One potential impact is through modification of aerosol surface tension. While atmospheric models typically assume a surface tension of 72 mN m−1, corresponding to pure water, PDMS can exhibit much lower values, down to 20 mN m−1 (Gaines, 1969; Kanellopoulos and Owen, 1971; Ananthapadmanabhan et al., 1990), similar to those of traffic-related lubricant oils (∼25 mN m−1) (Goebel and Lunkenheimer, 1997; Winoto et al., 2014). The interfacial tension between methylsiloxanes and water (30–50 mN m−1) (Kanellopoulos and Owen, 1971; El-Hamouz, 2007; Nowak et al., 2016) further supports the expectation that large molecular species may readily spread across droplet surfaces (Model S2 and Eq. S5) (Harkins, 1952; Winoto et al., 2014; Soloviev et al., 2016). Such spreading could substantially alter the interfacial properties of aerosols. Simple Köhler theory estimates (Equation S6) (Petters and Kreidenweis, 2007) suggest that, if sufficient large molecular methylsiloxanes coat aerosol surfaces, the critical supersaturation required for cloud droplet activation could be reduced by more than an order of magnitude. This magnitude of change is consistent with their industrial role as efficient defoaming agents, where trace additions strongly perturb fluid properties. However, direct experimental evidence for these atmospheric effects is currently lacking and should be pursued in future work.
Additionally, owing to the low freezing points, methylsiloxanes widely serve as constituents in anti-freeze formulations for the chemical industry and for cold-weather operations of vehicles and industrial equipment. Therefore, the presence of large molecular methylsiloxanes in atmospheric aerosols can potentially inhibit ice nucleation (IN), necessitating further investigation.
4.3 Implications
This study provides the first evidence that large molecular methylsiloxanes are widespread in atmospheric aerosols. As a newly recognized class of pollutants, their potential impacts on human health and the environment remain largely unknown, calling for urgent attention from both the scientific community and society at large. Substantial knowledge gaps exist, and systematic research is needed to address them. It should be noted that the samples analyzed here primarily originate from Europe and South America, and may not capture methylsiloxane variability in regions such as Asia, Africa, or North America. Additionally, Lithuania's small geographic size, with urban, coastal, and forested areas in close proximity, may result in overlapping influences from multiple emission sources and local meteorology.
Overall, the potential environmental implications of methylsiloxanes in atmospheric particulate matter can be summarized in three main aspects (Fig. 5). First, the inhalation of methylsiloxanes via particulate matter may pose potential health risks that require assessment. Second, the atmospheric fate of methylsiloxanes in particulate matter is not yet understood, including the processes of oxidation, fragmentation, hydrolysis, and other reactions occurring in both the gas and particle phase, as well as their impact on reactions of the organic and aqueous phases in aerosols. In addition, methylsiloxanes may alter the physical properties of aerosols, such as reducing surface tension (affecting their ability to act as cloud condensation nuclei) or exhibiting anti-freezing behavior (impacting ice nucleation). These possibilities also warrant further study. Third, methylsiloxanes are purely synthetic, relatively stable in atmospheric particulate matter, and emitted on a large scale, rendering them valuable indicators of anthropogenic emissions and atmospheric transport processes, especially in remote regions. Collectively, these considerations highlight the need for comprehensive and timely research on methylsiloxanes in atmospheric particulate matter, given their potential implications for human health, climate change, and environmental sustainability.
Our measurements reveal that large molecular methylsiloxanes are abundant in atmospheric aerosols from three countries representing diverse environments. Methylsiloxanes contribute up to 2.0 %–4.3 % of the total organic aerosol mass detected by thermal-desorption proton transfer reaction mass spectrometry using a maximum desorption temperature of 350 °C. Large molecular methylsiloxanes are detected via their characteristic fragmentation properties upon heating. The thermal-desorption profiles indicate that traffic emissions constitute a major source of large molecular methylsiloxanes across all ambient environments. Comparisons between tunnel and ambient air samples in São Paulo, Brazil, show that ambient air samples contain an increased fraction of methylsiloxanes of smaller molecular size at low desorption temperatures, presumably from gas-to-particle conversion processes. In the tunnel samples, methylsiloxanes and lubricating-oil hydrocarbon concentrations are highly correlated, indicating co-emission. During winter in Lithuania, methylsiloxanes and lubricating-oil hydrocarbons show a similar correlation in the ambient atmosphere, suggesting a common origin for both species and dilution as the major process during atmospheric transport. In São Paulo, methylsiloxane concentrations are similar in the tunnel and ambient atmosphere, whereas the concentration of lubricating-oil hydrocarbons decreases strongly in the ambient atmosphere. This shows the methylsiloxanes are chemically very stable and are likely to be transported over long distances. The estimated daily inhalation dose of methylsiloxanes via aerosols may be comparable to or exceed that of other synthetic compounds such as PFAS and MNPs, and studies assessing their toxicity are warranted. Their surface-tension-lowering and antifreezing properties may further influence the physical behavior and climatic effects of aerosols. Together, these findings highlight the ubiquity of this emerging pollutant and underscore the urgent need for systematic evaluation of its environmental and human health impacts.
Our research data has been included in the Supplement, as Supporting Data S1.
The supplement related to this article is available online at https://doi.org/10.5194/acp-26-5005-2026-supplement.
Conceptualization: PY, RH, UD. Methodology: PY, RH, UD. Investigation: PY, BSO, AM, HN. Visualization: PY, RH, UD. Supervision: RH, MFA, UD. Writing – original draft: PY, RH, UD. Writing – review & editing: PY, RH, BSO, AM, HN, DP, HNi, DM, MFA, RJH, UD.
The contact author has declared that none of the authors has any competing interests.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. The authors bear the ultimate responsibility for providing appropriate place names. Views expressed in the text are those of the authors and do not necessarily reflect the views of the publisher.
This study was funded by the Netherlands Organization for Scientific Research (NWO, grants nos. 820.01.001, and 834.08.002) and Foundation for Research Support of the São Paulo State (FAPESP; projects 2011/17754-2 and 2012/21456-0). This research has also been supported by the Cloud-Aerosol Interactions in a Nitrogen-dominated Atmosphere (CAINA) project (grant no. OCENW.XL21.XL21.112) and the Ruisdael Observatory co-financed by the Dutch Research Council (NWO, 184.034.015). Peng Yao would like to express appreciation for the support from China Scholarships Council (grant no. 201806320346).
This research has been supported by the Nederlandse Organisatie voor Wetenschappelijk Onderzoek (grant nos. 820.01.001 and 834.08.002), the Fundação de Amparo à Pesquisa do Estado de São Paulo (grant nos. 2011/17754-2 and 2012/21456-0), the Cloud-Aerosol Interactions in a Nitrogen-dominated Atmosphere (CAINA) project (grant no. OCENW.XL21.XL21.112), the Ruisdael Observatory co-financed by the Dutch Research Council (NWO, 184.034.015), and the China Scholarship Council (grant no. 201806320346).
This paper was edited by Arthur Chan and reviewed by Kangwei Li and one anonymous referee.
Allan, M. and Richardson, G. M.: Probability Density Functions Describing 24-Hour Inhalation Rates For Use in Human Health Risk Assessments, Hum. Ecol. Risk Assess. Int. J., 4, 379–408, https://doi.org/10.1080/10807039891284389, 1998.
Alton, M. W. and Browne, E. C.: Atmospheric Chemistry of Volatile Methyl Siloxanes: Kinetics and Products of Oxidation by OH Radicals and Cl Atoms, Environ. Sci. Technol., 54, 5992–5999, https://doi.org/10.1021/acs.est.0c01368, 2020.
Ananthapadmanabhan, K. P., Goddard, E. D., and Chandar, P.: A study of the solution, interfacial and wetting properties of silicone surfactants, Colloids Surf., 44, 281–297, https://doi.org/10.1016/0166-6622(90)80202-F, 1990.
Brunet, C. E., Marek, R. F., Stanier, C. O., and Hornbuckle, K. C.: Concentrations of Volatile Methyl Siloxanes in New York City Reflect Emissions from Personal Care and Industrial Use, Environ. Sci. Technol., 58, 8835–8845, https://doi.org/10.1021/acs.est.3c10752, 2024.
Bzdek, B. R., Horan, A. J., Pennington, M. R., Janechek, N. J., Baek, J., Stanier, C. O., and Johnston, M. V.: Silicon is a Frequent Component of Atmospheric Nanoparticles, Environ. Sci. Technol., 48, 11137–11145, https://doi.org/10.1021/es5026933, 2014.
Camino, G., Lomakin, S. M., and Lazzari, M.: Polydimethylsiloxane thermal degradation Part 1. Kinetic aspects, Polymer (Guildf.), 42, 2395–2402, https://doi.org/10.1016/S0032-3861(00)00652-2, 2001.
Claight Corporation: Silicones and siloxanes market size, share and outlook – growth analysis report and forecast trends (2026–2035), https://www.expertmarketresearch.com/reports/silicones-and-siloxanes-market, last access: 17 February 2026.
Coggon, M. M., McDonald, B. C., Vlasenko, A., Veres, P. R., Bernard, F., Koss, A. R., Yuan, B., Gilman, J. B., Peischl, J., Aikin, K. C., DuRant, J., Warneke, C., Li, S.-M., and de Gouw, J. A.: Diurnal Variability and Emission Pattern of Decamethylcyclopentasiloxane (D5) from the Application of Personal Care Products in Two North American Cities, Environ. Sci. Technol., 52, 5610–5618, https://doi.org/10.1021/acs.est.8b00506, 2018.
Decker, Z. C. J., Alpert, P. A., Ammann, M., Anet, J. G., Bauer, M., Cui, T., Durdina, L., Edebeli, J., Gysel-Beer, M., Prévôt, A. S. H., Qi, L., Slowik, J. G., Spirig, C., Tinorua, S., Ungeheuer, F., Vogel, A., Zhang, J., and Brem, B. T.: Emission and Formation of Aircraft Engine Oil Ultrafine Particles, ACS ES&T Air, 12, 1662–1672, https://doi.org/10.1021/acsestair.4c00184, 2024.
Dusek, U., Hitzenberger, R., Kasper-Giebl, A., Kistler, M., Meijer, H. A. J., Szidat, S., Wacker, L., Holzinger, R., and Röckmann, T.: Sources and formation mechanisms of carbonaceous aerosol at a regional background site in the Netherlands: insights from a year-long radiocarbon study, Atmos. Chem. Phys., 17, 3233–3251, https://doi.org/10.5194/acp-17-3233-2017, 2017.
Eichler, P., Müller, M., Rohmann, C., Stengel, B., Orasche, J., Zimmermann, R., and Wisthaler, A.: Lubricating Oil as a Major Constituent of Ship Exhaust Particles, Environ. Sci. Technol. Lett., 4, 54–58, https://doi.org/10.1021/acs.estlett.6b00488, 2017.
El-Hamouz, A.: Effect of Surfactant Concentration and Operating Temperature on the Drop Size Distribution of Silicon Oil Water Dispersion, J. Dispers. Sci. Technol., 28, 797–804, https://doi.org/10.1080/01932690701345893, 2007.
European Chemicals Agency (ECHA): Candidate List of Substances of Very High Concern for Authorisation, https://echa.europa.eu/candidate-list-table (last access: 8 March 2026), 2024.
Evich, M. G., Davis, M. J. B., McCord, J. P., Acrey, B., Awkerman, J. A., Knappe, D. R. U., Lindstrom, A. B., Speth, T. F., Tebes-Stevens, C., Strynar, M. J., Wang, Z., Weber, E. J., Henderson, W. M., and Washington, J. W.: Per- and polyfluoroalkyl substances in the environment, Science, 375, https://doi.org/10.1126/science.abg9065, 2022.
Faust, J. A.: PFAS on atmospheric aerosol particles: a review, Environ. Sci. Process. Imp., 25, 133–150, https://doi.org/10.1039/D2EM00002D, 2023.
Franzen, A., Van Landingham, C., Greene, T., Plotzke, K., and Gentry, R.: A global human health risk assessment for Decamethylcyclopentasiloxane (D5), Regul. Toxicol. Pharmacol., 74, S25–S43, https://doi.org/10.1016/j.yrtph.2015.10.023, 2016.
Franzen, A., Greene, T., Van Landingham, C., and Gentry, R.: Toxicology of octamethylcyclotetrasiloxane (D4), Toxicol. Lett., 279, 2–22, https://doi.org/10.1016/j.toxlet.2017.06.007, 2017.
Gaines, G. L.: Surface tension of polymer solutions. I. Solutions of poly(dimethylsiloxanes), J. Phys. Chem., 73, 3143–3150, https://doi.org/10.1021/j100843a060, 1969.
Gentry, R., Franzen, A., Van Landingham, C., Greene, T., and Plotzke, K.: A global human health risk assessment for octamethylcyclotetrasiloxane (D4), Toxicol. Lett., 279, 23–41, https://doi.org/10.1016/j.toxlet.2017.05.019, 2017.
Genualdi, S., Harner, T., Cheng, Y., MacLeod, M., Hansen, K. M., van Egmond, R., Shoeib, M., and Lee, S. C.: Global Distribution of Linear and Cyclic Volatile Methyl Siloxanes in Air, Environ. Sci. Technol., 45, 3349–3354, https://doi.org/10.1021/es200301j, 2011.
Glüge, J., Scheringer, M., Cousins, I. T., DeWitt, J. C., Goldenman, G., Herzke, D., Lohmann, R., Ng, C. A., Trier, X., and Wang, Z.: An overview of the uses of per- and polyfluoroalkyl substances (PFAS), Environ. Sci. Process. Imp., 22, 2345–2373, https://doi.org/10.1039/D0EM00291G, 2020.
Goebel, A. and Lunkenheimer, K.: Interfacial Tension of the Water/ n -Alkane Interface, Langmuir, 13, 369–372, https://doi.org/10.1021/la960800g, 1997.
Han, C., Yang, H., Li, K., Lee, P., Liggio, J., Leithead, A., and Li, S.-M.: Secondary organic aerosols from OH oxidation of cyclic volatile methyl siloxanes as an important Si source in the atmosphere, Atmos. Chem. Phys., 22, 10827–10839, https://doi.org/10.5194/acp-22-10827-2022, 2022.
Harkins, W. D.: The physical chemistry of surface films, Reinhold, https://cir.nii.ac.jp/crid/1971430859792846500 (last access: 17 February 2026), 1952.
Holzinger, R.: PTRwid: A new widget tool for processing PTR-TOF-MS data, Atmos. Meas. Tech., 8, 3903–3922, https://doi.org/10.5194/amt-8-3903-2015, 2015.
Holzinger, R., Williams, J., Herrmann, F., Lelieveld, J., Donahue, N. M., and Röckmann, T.: Aerosol analysis using a Thermal-Desorption Proton-Transfer-Reaction Mass Spectrometer (TD-PTR-MS): a new approach to study processing of organic aerosols, Atmos. Chem. Phys., 10, 2257–2267, https://doi.org/10.5194/acp-10-2257-2010, 2010a.
Holzinger, R., Kasper-Giebl, A., Staudinger, M., Schauer, G., and Röckmann, T.: Analysis of the chemical composition of organic aerosol at the Mt. Sonnblick observatory using a novel high mass resolution thermal-desorption proton-transfer-reaction mass-spectrometer (hr-TD-PTR-MS), Atmos. Chem. Phys., 10, 10111–10128, https://doi.org/10.5194/acp-10-10111-2010, 2010b.
Holzinger, R., Acton, W. J. F., Bloss, W. J., Breitenlechner, M., Crilley, L. R., Dusanter, S., Gonin, M., Gros, V., Keutsch, F. N., Kiendler-Scharr, A., Kramer, L. J., Krechmer, J. E., Languille, B., Locoge, N., Lopez-Hilfiker, F., Materić, D., Moreno, S., Nemitz, E., Quéléver, L. L. J., Sarda Esteve, R., Sauvage, S., Schallhart, S., Sommariva, R., Tillmann, R., Wedel, S., Worton, D. R., Xu, K., and Zaytsev, A.: Validity and limitations of simple reaction kinetics to calculate concentrations of organic compounds from ion counts in PTR-MS, Atmos. Meas. Tech., 12, 6193–6208, https://doi.org/10.5194/amt-12-6193-2019, 2019.
Jambeck, J. R., Geyer, R., Wilcox, C., Siegler, T. R., Perryman, M., Andrady, A., Narayan, R., and Law, K. L.: Plastic waste inputs from land into the ocean, Science, 347, 768–771, https://doi.org/10.1126/science.1260352, 2015.
Janechek, N. J., Marek, R. F., Bryngelson, N., Singh, A., Bullard, R. L., Brune, W. H., and Stanier, C. O.: Physical properties of secondary photochemical aerosol from OH oxidation of a cyclic siloxane, Atmos. Chem. Phys., 19, 1649–1664, https://doi.org/10.5194/acp-19-1649-2019, 2019.
Kanellopoulos, A. G. and Owen, M. J.: Adsorption of sodium dodecyl sulphate at the silicone fluid/water interface, Trans. Faraday Soc., 67, 3127, https://doi.org/10.1039/tf9716703127, 1971.
Liu, X., Henzing, B., Hensen, A., Mulder, J., Yao, P., van Dinther, D., van Bronckhorst, J., Huang, R., and Dusek, U.: Measurement report: Evaluation of the TOF-ACSM-CV for PM1.0 and PM2.5 measurements during the RITA-2021 field campaign, Atmos. Chem. Phys., 24, 3405–3420, https://doi.org/10.5194/acp-24-3405-2024, 2024.
Masalaite, A., Holzinger, R., Remeikis, V., Röckmann, T., and Dusek, U.: Characteristics, sources and evolution of fine aerosol (PM1) at urban, coastal and forest background sites in Lithuania, Atmos. Environ., 148, 62–76, https://doi.org/10.1016/j.atmosenv.2016.10.038, 2017.
Masalaite, A., Holzinger, R., Ceburnis, D., Remeikis, V., Ulevičius, V., Röckmann, T., and Dusek, U.: Sources and atmospheric processing of size segregated aerosol particles revealed by stable carbon isotope ratios and chemical speciation, Environ. Pollut., 240, 286–296, https://doi.org/10.1016/j.envpol.2018.04.073, 2018.
Materić, D., Peacock, M., Kent, M., Cook, S., Gauci, V., Röckmann, T., and Holzinger, R.: Characterisation of the semi-volatile component of Dissolved Organic Matter by Thermal Desorption – Proton Transfer Reaction – Mass Spectrometry, Sci. Rep., 7, 15936, https://doi.org/10.1038/s41598-017-16256-x, 2017.
Mohamed Nor, N. H., Kooi, M., Diepens, N. J., and Koelmans, A. A.: Lifetime Accumulation of Microplastic in Children and Adults, Environ. Sci. Technol., 55, 5084–5096, https://doi.org/10.1021/acs.est.0c07384, 2021.
Nowak, E., Kovalchuk, N. M., Che, Z., and Simmons, M. J. H.: Effect of surfactant concentration and viscosity of outer phase during the coalescence of a surfactant-laden drop with a surfactant-free drop, Colloids Surf. A, 505, 124–131, https://doi.org/10.1016/j.colsurfa.2016.02.016, 2016.
Oyama, B. S., Andrade, M. D. F., Herckes, P., Dusek, U., Röckmann, T., and Holzinger, R.: Chemical characterization of organic particulate matter from on-road traffic in São Paulo, Brazil, Atmos. Chem. Phys., 16, 14397–14408, https://doi.org/10.5194/acp-16-14397-2016, 2016.
Petters, M. D. and Kreidenweis, S. M.: A single parameter representation of hygroscopic growth and cloud condensation nucleus activity, Atmos. Chem. Phys., 7, 1961–1971, https://doi.org/10.5194/acp-7-1961-2007, 2007.
PlasticsEurope: Plastics – the Facts 2022, 81 pp., https://plasticseurope.org/knowledge-hub/plastics-the-facts-2022 (last access: 17 February 2026), 2022.
Quinn, A. L., Regan, J. M., Tobin, J. M., Marinik, B. J., McMahon, J. M., McNett, D. A., Sushynski, C. M., Crofoot, S. D., Jean, P. A., and Plotzke, K. P.: In Vitro and In Vivo Evaluation of the Estrogenic, Androgenic, and Progestagenic Potential of Two Cyclic Siloxanes, Toxicol. Sci., 96, 145–153, https://doi.org/10.1093/toxsci/kfl185, 2006.
Revell, L. E., Kuma, P., Le Ru, E. C., Somerville, W. R. C., and Gaw, S.: Direct radiative effects of airborne microplastics, Nature, 598, 462–467, https://doi.org/10.1038/s41586-021-03864-x, 2021.
Rücker, C. and Kümmerer, K.: Environmental Chemistry of Organosiloxanes, Chem. Rev., 115, 466–524, https://doi.org/10.1021/cr500319v, 2015.
Soloviev, A. V., Haus, B. K., McGauley, M. G., Dean, C. W., Ortiz-Suslow, D. G., Laxague, N. J. M., and Özgökmen, T. M.: Surface dynamics of crude and weathered oil in the presence of dispersants: Laboratory experiment and numerical simulation, J. Geophys. Res.-Oceans, 121, 3502–3516, https://doi.org/10.1002/2015JC011533, 2016.
Sonntag, D. B., Bailey, C. R., Fulper, C. R., and Baldauf, R. W.: Contribution of Lubricating Oil to Particulate Matter Emissions from Light-Duty Gasoline Vehicles in Kansas City, Environ. Sci. Technol., 46, 4191–4199, https://doi.org/10.1021/es203747f, 2012.
Stifelman, M.: Using doubly-labeled water measurements of human energy expenditure to estimate inhalation rates, Sci. Total Environ., 373, 585–590, https://doi.org/10.1016/j.scitotenv.2006.11.041, 2007.
Tang, X., Misztal, P. K., Nazaroff, W. W., and Goldstein, A. H.: Siloxanes Are the Most Abundant Volatile Organic Compound Emitted from Engineering Students in a Classroom, Environ. Sci. Technol. Lett., 2, 303–307, https://doi.org/10.1021/acs.estlett.5b00256, 2015.
Theodosi, C., Grivas, G., Zarmpas, P., Chaloulakou, A., and Mihalopoulos, N.: Mass and chemical composition of size-segregated aerosols (PM1, PM2.5, PM10) over Athens, Greece: local versus regional sources, Atmos. Chem. Phys., 11, 11895–11911, https://doi.org/10.5194/acp-11-11895-2011, 2011.
Thomas, T. H. and Kendrick, T. C.: Thermal analysis of polydimethylsiloxanes. I. Thermal degradation in controlled atmospheres, J. Polym. Sci. Pt. A-2, 7, 537–549, https://doi.org/10.1002/pol.1969.160070308, 1969.
Timkovsky, J., Dusek, U., Henzing, J. S., Kuipers, T. L., Röckmann, T., and Holzinger, R.: Offline thermal-desorption proton-transfer-reaction mass spectrometry to study composition of organic aerosol, J. Aerosol Sci., 79, 1–14, https://doi.org/10.1016/j.jaerosci.2014.08.010, 2015.
US EPA – United States Environmental Protection Agency: Final Scope of the Risk Evaluation for Octamethylcyclotetra-siloxane, https://www.epa.gov/system/files/documents/2022-03/casrn_556_67_2-octamethylcyclotetra-siloxane-d4_finalscope.pdf (last access: 17 February 2026), 2022.
Winoto, Loahardjo, N., Takamura, K., and Morrow, N. R.: Spreading and Retraction of Spilled Crude Oil on Seawater, Int. Oil Spill Conf. Proc., 2014, 1465–1473, https://doi.org/10.7901/2169-3358-2014.1.1465, 2014.
Worton, D. R., Moreno, S., O'Daly, K., and Holzinger, R.: Development of an International System of Units (SI)-traceable transmission curve reference material to improve the quantitation and comparability of proton-transfer-reaction mass-spectrometry measurements, Atmos. Meas. Tech., 16, 1061–1072, https://doi.org/10.5194/amt-16-1061-2023, 2023.
Yao, P., Chianese, E., Kairys, N., Holzinger, R., Materić, D., Sirignano, C., Riccio, A., Ni, H., Huang, R.-J., and Dusek, U.: A large contribution of methylsiloxanes to particulate matter from ship emissions, Environ. Int., 165, 107324, https://doi.org/10.1016/j.envint.2022.107324, 2022.
Yao, P., Holzinger, R., Materić, D., Oyama, B. S., de Fátima Andrade, M., Paul, D., Ni, H., Noto, H., Huang, R.-J., and Dusek, U.: Methylsiloxanes from Vehicle Emissions Detected in Aerosol Particles, Environ. Sci. Technol., 57, 14269–14279, https://doi.org/10.1021/acs.est.3c03797, 2023.
Yucuis, R. A., Stanier, C. O., and Hornbuckle, K. C.: Cyclic siloxanes in air, including identification of high levels in Chicago and distinct diurnal variation, Chemosphere, 92, 905–910, https://doi.org/10.1016/j.chemosphere.2013.02.051, 2013.