the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Organic acids and cloud droplet acidity in recent years at Whiteface Mountain, NY, with a focus on wildfire smoke influence
Archana Tripathy
Haider A. Khwaja
Mirza M. Hussain
Elizabeth Yerger
Daniel Kelting
Christopher E. Lawrence
Paul Casson
Phil Snyder
Sara Lombardo
Noah Pittman
Kathleen DeMarle
Rudra Patel
Lily Hammond
Eric C. Apel
Rebecca S. Hornbrook
Alan J. Hills
Richard Brandt
Scott McKim
Jim Schlemmer
Clouds facilitate the transformation of atmospheric gases and particles, yet the impact of cloud processing on organic compounds remains poorly understood due to the paucity of routine measurements within aqueous samples (e.g., cloud water and precipitation). This study presents seven years (2018–2024) of routine summertime measurements of three major Low Molecular Weight Organic Acids (LMWOA: formate, acetate and oxalate) in cloud water samples collected from the summit of Whiteface Mountain in the northeastern United States, including their contributions to Dissolved Organic Carbon (DOC), ion balance, and cloud water acidity, with critical evaluation of sample handling procedures to minimize volatilization, microbial degradation and contamination. Formate and acetate were the dominant monocarboxylic acids, exhibiting seasonal variability consistent with changes in biogenic emissions, whereas oxalate showed higher concentrations in smoke-impacted clouds. A growing fraction of samples exhibit surplus ammonium relative to sulfate and nitrate concentrations, which previous research hypothesized results from unmeasured organic acids, consistent with the positive relationship between surplus ammonium and LMWOA concentrations observed in the current study. An observed correlation between oxalate and DOC, with higher slope at higher ozone concentrations, supports enhanced in-cloud secondary production of oxalate under high oxidant levels. A comparison of “Aged” versus “Fresh” wildfire smoke-influenced samples suggest that aging can enhance ammonium, with heavier organic acids dominating the DOC pool and acidity, whereas fresh plumes were primarily influenced by directly emitted LMWOA. This study highlights the need for continued monitoring of the evolving cloud water chemistry to better understand the broader impacts on atmospheric chemistry.
- Article
(1651 KB) - Full-text XML
-
Supplement
(1538 KB) - BibTeX
- EndNote
Cloud water plays a major role in atmospheric chemistry by providing a medium for aqueous phase chemical reactions that transform the chemical composition of particles and gases, contributing to the formation of secondary organic aerosols (SOA) (Ervens et al., 2014). These processes can alter aerosol chemistry, cloud microphysics and the Earth’s radiation budget. Aqueous processing of organics within aerosol liquid water has also been shown to enhance SOA formation under humid conditions in urban field studies (Duan et al., 2021; Rogers et al., 2025). While gas phase chemistry dominates in clear sky conditions, clouds enable unique multiphase reactions where gases and particles can get scavenged, dissolved, and interact (Barth et al., 2000). In-cloud oxidation of sulfur dioxide and nitrogen oxide into sulfate and nitrate leads to acid rain formation, damaging regional air quality and ecosystem (Husain et al., 1991; Rattigan et al., 2001).
Inorganic compounds of cloud water such as sulfate, nitrate, and ammonium have been extensively studied due to their contribution to acid rain and haze formation (Hayden et al., 2008; Hand et al., 2012; Murray et al., 2013). In contrast, organic compounds remain less understood. Total organic carbon (TOC) is often used as a bulk measurement of organic compounds in cloud water. The CPOC pilot study from Whiteface Mountain (WFM) in 2017 revealed that organic matter dominates the chemical composition of particulate matter (Zhang et al., 2019), consistent with evidence from another remote mountaintop atmospheric research station at Puy de Dôme (PUY) France (Chen et al., 2022), a key long-term measurement site in Europe where cloud processes and atmospheric composition are studied (Baray et al., 2020). The CPOC pilot study also established that long-range transported wildfire smoke was efficiently scavenged by clouds at WFM, highlighting the critical role of aqueous processes in shaping both aerosol and cloud water chemistry (Lance et al., 2020). Lee et al. (2022) assessed the influence of wildfire smoke on TOC concentrations at WFM. However, their results were based on a subset of cloud water samples that fulfilled a traditional inorganic ion balance criteria, thereby ignoring a major fraction of samples with elevated TOC levels, as discussed in a subsequent study that showed TOC correlates strongly with inorganic ion imbalance (Lawrence et al., 2023). This long-term cloud water observation study from WFM also reported an increasing trend in TOC concentrations over the past decade (Lawrence et al., 2023). A similar trend in dissolved organic matter has been reported in precipitation samples collected at Hubbard Brook Experimental Forest, another northeastern U.S. site (Green et al., 2025). This rising organic carbon trend may be driven by a combination of factors such as increasing local biogenic emissions from forest photosynthesis or heat stress responses, increasing prevalence of wildfire smoke events, shifts in atmospheric oxidation chemistry due to declining NOx, or a shift in cloud pH following implementation of the Clean Air Act (CAA), which could promote the partitioning of organic compounds into the aqueous phase (Lawrence et al., 2023; Lance et al., 2025; Green et al., 2025). Studies from PUY indicate that TOC concentrations in cloud water can vary widely, with a significant contribution from pollution driven by anthropogenic emissions (Deguillaume et al., 2014). Similar chemical variability in cloud and fog water composition has also been observed at other mountaintop sites in the Swiss Alps (Michna et al., 2015), Mt. Brocken in Germany (Plessow et al., 2001), Mt. Taishan (Wang et al., 2011) and Mt. Lu (Sun et al., 2015) in China, all of which show strong influence from long-range transported pollution, dust and regional emissions. However, TOC measurements alone are insufficient for understanding the source, transformation, and chemical reactivity of organic compounds in cloud water (van Pinxteren et al., 2016). Molecular-level analysis of organic species is necessary to address the diversity of this huge fraction of organic compounds undergoing secondary processing in the aqueous phase and to understand the partitioning between dissolved and particulate phases (Lim et al., 2010; Herckes et al., 2013).
Among the diverse organic species in cloud water, organic acids represent a dominant and chemically reactive subset (Khwaja, 1995; Marinoni et al., 2004; van Pinxteren et al., 2016). The C1–C4 monocarboxylic (formic, acetic, pyruvic, lactic, glyoxylic, butyric acids) and dicarboxylic (oxalic, malonic, succinic, malic and glutaric acids) organic acids represent a group of low molecular weight organic acids identified in cloud water by several studies as discussed below, whereas C6 or higher are considered higher molecular weight organic acids. Photochemical oxidation of Biogenic Volatile Organic Compounds (BVOC) significantly contributes to the formation of formic and acetic acids in the aqueous phase (Chebbi and Carlier, 1996). Oxalic acid has been shown to be the most abundant dicarboxylic acid, followed by malonic and succinic acids, which are primarily formed through photochemical reactions from both natural and anthropogenic sources (Grosjean et al., 1978; Hatakeyama et al., 1987; Kawamura and Ikushima, 1993; Khwaja, 1995; Cong et al., 2015; Kawamura and Bikkina, 2016). In a seminal 1995 study at WFM, low molecular weight organic acids accounted for 22 % of acidity and 7 %–15 % of Dissolved Organic Carbon (DOC) (Khwaja et al., 1995), and organic acids have previously been shown to contribute more than 20 % to cloud water ionic content at PUY (Marinoni et al., 2004). Secondary processes, including aqueous phase transformation such as oxidation, hydration, metal-catalyzed reaction, and oligomerization of precursors like glyoxal and methylglyoxal formed by gas phase oxidation, lead to the formation of di- and oxy-carboxylic acids in cloud droplets (Carlton et al., 2007; Altieri et al., 2008; Yang et al., 2008; Tan et al., 2010). The complex interplay of chemical reactions and atmospheric conditions during long-range transport influences the partitioning of organic acids (Zhao and Gao, 2008; Carlton et al., 2009; Paulot et al., 2011; Eugene et al., 2018).
Organic acids have been measured for short durations in cloud and fog water samples from high elevation remote sites, such as PUY (Deguillaume et al., 2014; van Pinxteren et al., 2016), WFM (Khwaja et al., 1995; Sagona et al., 2014; Lee et al., 2022; Cook et al., 2017), East peak in Puerto Rico (Gioda et al., 2011), Mt. Fuji in Japan (Kunwar et al., 2019), Mt. Hua in China (Shen et al., 2024) and Mt. Tai Mo Shan in Hong Kong (Zhao et al., 2019; Li et al., 2020). However, routine monitoring of organic acids remains a challenge due to their chemical instability, low concentrations, volatility, and possible microbial degradation, in addition to analytical limitations like co-elution with inorganic and other organic analytes. Sampling conditions such as temperature may also influence organic acid levels via: (1) Microbial degradation (Amato et al., 2007; Nuñez López et al., 2024), and (2) volatilization of some organic acids (Tao and Murphy, 2019), especially at low cloud water pH. Hence, meticulous handling of samples is needed from the time of collection to analysis, making it challenging to add organic acids to the suite of routine measurements. Despite the many associated challenges, studies have emphasized the need for additional cloud water organic acid measurements, to better constrain the multiphase chemical processes driving aqueous SOA formation and aging (Ervens et al., 2011; Hennigan et al., 2015; Barth et al., 2021; Liu et al., 2021).
Growing evidence suggests that aqueous organic and reactive nitrogen chemistry are linked (Xu et al., 2020; Lv et al., 2022). Organic acids influence partitioning and aerosol acidity (Hennigan et al., 2015; Tao and Murphy, 2019), as well as pH buffering in moderately acidic droplets (Zheng et al., 2023b). Nitrogen-containing organic compounds comprise a major fraction of dissolved organic matter in cloud water and precipitation samples (Cook et al., 2017; Bianco et al., 2019; Murray et al., 2022), with as much as half of detected organic molecules classified as CHON within cloud water samples at a mountain top site in southeastern China (Shu et al., 2025). Enrichment of CHN and CHON compounds has been directly linked to aqueous processing in several studies (Boone et al., 2015; Kim et al., 2019; Mattsson et al., 2025; Kuang et al., 2025; Jin et al., 2026). Lab studies have shown that dissolved NH3 may react with carbonyls to form nitrogen-containing organic compounds like imidazoles, with faster production observed in small, concentrated and/or evaporating droplets (De Haan et al., 2011; Lee et al., 2013; De Haan et al., 2020; Marracci and Murray, 2025). However, Boreal wildfire smoke plumes have also shown increased contributions of CHON compounds with aging (Ditto et al., 2021), presumably in the absence of clouds (though relative humidity and aerosol liquid water content were not discussed in that paper). Likewise, organo-nitrogen production has been observed in chamber experiments held at 50 % relative humidity (Liu et al., 2015), and nitrate radical BVOC oxidation can be an efficient nighttime source of organonitrates under cloud-free conditions (Ng et al., 2017; Slade et al., 2019), indicating that aqueous processes may not always be required for secondary formation of CHON compounds. Thus, the role of reactive nitrogen in aqueous SOA formation can be complex, potentially involving competing multiphase mechanisms, and different outcomes may be expected for clouds influenced by wildfire smoke, agricultural emissions and/or varying mixtures of biogenic and anthropogenic precursors.
In cloud water at WFM, substantial long-term changes in both organic and inorganic solute concentrations have occurred simultaneously. The strong correlation between TOC and inorganic ion imbalance suggests that organic acids comprise a major and growing fraction of anions (Lawrence et al., 2023), highlighting a critical need for long-term measurements of organic acids alongside measurements of inorganic ions. The only previous published measurements of formic, acetic, and oxalic acids at WFM come from three cloud events sampled in summer 1987 (Khwaja et al., 1995), leaving a critical gap in understanding their role in cloud chemistry under the new chemical regime post-CAA. This lack of long-term organic acid datasets limits models from accurately predicting their concentrations. Current simulations tend to underestimate organic acid levels (Barth et al., 2021; Lawrence et al., 2024), especially in polluted conditions, suggesting that key factors controlling essential sources, formation pathways, and removal processes of organic acids are inadequately represented in model parameterizations.
This study presents a seven-year study of summertime cloud water formate, acetate, and oxalate concentrations (the deprotonated ions of formic, acetic, and oxalic acids), representing the first routine long-term measurements of Low Molecular Weight Organic Acids (LMWOA) in cloud water at a remote high- elevation site, to our knowledge. The semi-automated cloud water collection system used in this study significantly increases the capacity of cloud events sampled, in contrast to an intensive previous study of LMWOA in cloud water from this site for three cloud events in 1987 (Khwaja et al., 1995). Hence, this study offers deeper insight into seasonal patterns and episodic wildfire smoke events influencing cloud water composition, especially LMWOA concentrations, addressing an important knowledge gap.
2.1 Site Description
Whiteface Mountain (WFM) Atmospheric Research Observatory, located in the remote high peaks regions of upstate New York, northeastern United States, is an exceptional site for investigating cloud water chemistry. It is situated at an elevation of 1483 m (4867 ft) and spends 20 %–60 % of the time during summer immersed in clouds (Schwab et al., 2016). This mountain top site is the 5th highest peak of the Adirondack Mountains and downwind of diverse natural and anthropogenic emission sources from Canada, the Great Lakes, and the western United States. Since the 1970s, WFM has been providing critical insights into atmospheric chemistry, influencing CAA regulatory actions in the 1990s aimed at reducing acid deposition (Falconer and Falconer, 1980; Mohnen and Vong, 1993; Dukett et al., 2011).
Spanning from June through September each year, the dataset captures warm cloud events during the late spring (June), summer (July–August), and early fall (September). These variations are influenced by biogenic activity during the plant growth season in the late spring and meteorological shifts across the sampling period. Additionally, the dataset includes periods of episodic wildfire events, which commonly occur during the summer months. Fall season is also influenced by biogenic emissions due to leaf foliage. By including both regular seasonal cycles and intermittent high-impact smoke as well as heatwave events, this study offers a comprehensive view of the factors influencing LMWOA dynamics in cloud water chemistry. Furthermore, the remote location of WFM minimizes direct anthropogenic influence, with minimal local pollution interference, enabling the observation of broader atmospheric trends. This makes WFM highly relevant for studying the transport and transformation of air masses influenced by regional and long-range emissions, making it a one-of-a-kind high-elevation site to investigate cloud water chemistry. It should be noted that this study does not include wintertime measurements due to operational limitations at the site during freezing conditions from late fall to May. Sampling ends by the first week of October to prevent problems associated with riming. Therefore, the dataset focuses exclusively on warm cloud measurements, capturing the most dynamic period of cloud water chemistry.
2.2 Sample collection methods and offline chemical analyses
Cloud water samples were collected at WFM using an automated Mohnen Omni-directional passive cloud water collector equipped with Teflon strings (0.035–0.04 mm diameter) with a 50 % droplet cut-off diameter of 2–5 µm at typical summit wind speeds of 2–10 m s−1 (Mohnen, 1979; Mohnen and Kadlecek, 1989). Sampling occured under specific meteorological conditions: Liquid Water Content (LWC) >0.05 g m−3 (measured by a Gerber Particle Volume Monitor), temperature >2 °C, no rain (detected by a CAPMoN rain sensor), and wind speed >2 m s−1 (measured by an RM Young anemometer). When these conditions were met for at least one minute, the collector exposed Teflon strings to capture cloud water from the passing airflow (Lawrence et al., 2023). Samples were collected every 12 h into a refrigerated 1 L ISCO bottle connected to a 12 L high-density polyethylene (HDPE) accumulator vessel that is continuously weighed to monitor accumulated cloud water sample volume. The collected water was filtered through an in-line 0.4 µm track-etched polycarbonate filter using a peristaltic pump to prevent microbial degradation, and filters were replaced twice a week. From 16 July 2024, we started using 0.2 µm filters for microbial analysis (Lombardo et al., 2025). Each sample was then transferred to a 250 mL bottle and kept frozen until chemical analysis. While LMWOA measurements have been reported for specific cloud events at various locations, including WFM, routine bulk sampling at a remote site required careful sample handling to prevent degradation or volatile losses. All samples were filtered during collection and stored at 2 °C to prevent microbial degradation during the three-day holding time until delivery to the lab for chemical analysis. Chloroform (CHCl3) was not added to the cloud water samples since controlled administration of CHCl3 concentrations based on collected sample volume is not yet a feature of the automated cloud water collection system. It should be noted that Khwaja et al. (1995) used CHCl3 and 0.4 µm filters prior to organic acid analysis of cloud water samples collected on an hourly basis for a short-term study at WFM in 1987. Although 0.2 µm filters are widely regarded as sufficient for sterilization purposes, Hahn (2004) isolated a wide variety of viable bacteria from 0.2 µm filtered freshwater samples, suggesting that even with this level of filtration, some microbes may end up in the filtered cloud water samples. Routine blank and rinse samples of the cloud water collection system using Type 1 ultrapure water (18 MΩ cm resistivity) were collected multiple times each year to assess for contamination. A table showing measurement statistics for these samples is added to the Supplement (Table S3).
Detailed descriptions of the measurement methods for each year and each analyte are presented in the Supplement following some of the procedures described in previous studies (Khwaja et al., 1999; Tripathy et al., 2024; Lawrence et al., 2023). In each year of the study, quality control samples were prepared fresh daily and monitored to assess column performance. Lab duplicates were analyzed by randomly selecting one sample from that day's batch. Results within a relative percent difference (RPD) acceptance criterion of ±10 % were accepted; otherwise, more duplicates were analyzed or the entire batch was reanalyzed.
This study presents a seven-year study of three major LMWOA (formate, acetate, and oxalate). Although acetate was suspected to co-elute with glycolate in a few instances, the peaks for acetate were resolvable, and glycolate concentrations were consistently below the detection limit. Additionally, measurements for other organic acids were limited due to analytical limitations. Hence, the focus of this paper is on the three most abundant LMWOA. Method detection limits (MDL) for each organic acid analyte are provided in Table S1, along with the corresponding laboratories where the measurements were conducted for each year that samples were collected. More details about methods for inorganic analytes can be found in Lawrence et al. (2023).
Analyte concentrations measured by ion chromatography (mg L−1) are converted to mol L−1 by dividing by molar mass and then converted to equivalent concentrations (eq L−1) by multiplying by ionic charge. The total concentration of measured cations (∑Cat) and anions (∑An) are then calculated according to the following equations, with concentrations for all analytes in units of eq L−1:
where ∑LMWOA is the sum of the measured formate, acetate, and oxalate concentrations. It should be noted that these are the maximum possible contributions of these LMWOA to the ion balance since these values do not account for the deprotonation state of those anions (except as calculated in Eqs. 5 and 6, and reported in Figs. 7–9).
Then we infer the concentrations of missing anions by assuming that electrochemical charge balance was sustained in these bulk aqueous solutions, but assuming that the anions listed in Eq. (2) were insufficient to characterize all the anions present. Inherent to Eqs. (3) and (4) is the assumption that any cations other than those listed in Eq. (1) are present in negligible concentrations (Fig. S7 in the Supplement).
The measured ionic concentrations of each analyte are then divided by the total ion concentration (∑Ions) to determine ionic fractions, and charge imbalance is calculated from
Following the procedure of Lawrence et al. (2023), we estimate acidity for the vast majority of cloud droplets as they existed in the atmosphere prior to collection using a “top down” approach based on the measured bulk cloud water acidity and measured concentrations of the two dominant non-volatile cations (Mg2+ and Ca2+), which are presumed to exist entirely within the coarse mode aerosol population, therefore representing a very small fraction of the particles present even when comprising a significant fraction of the aerosol mass loading, i.e. , which is subsequently used to calculate pHTD. Similarly, a “bottom up” approach is used to estimate cloud droplet acidity based on measured concentrations of the dominant inorganic ions typically associated with the fine mode aerosol population, i.e., .
Since organic acids are also expected to exist primarily within the fine mode aerosol population (VandenBoer et al., 2011; Lawrence et al., 2023), in this study, we extend the “bottom up” acidity calculation to include the measured LMWOA concentrations, thereby introducing a new variable:
where , representing the dissociated fractions of the three LMWOAs calculated from their measured concentrations and pKa values at pHTD (our best estimate for cloud droplet pH), using equations from Seinfeld and Pandis (2016) (used in Figs. 7–9).
Similarly, the Missing Acid Fraction (MAF) concept introduced by Lawrence et al. (2023) was modified to include LMWOA, and the new variable MAF* is introduced here:
For this study, negative values for [H+]BU and [H+]BU* are set to zero for calculating pHBU and pHBU*, respectively, as well as for calculating MAF and MAF*.
Isoprene, a major BVOC at the site, was measured from grab samples collected in stainless steel whole air canisters from the base of WFM (610 m elevation) in the summer of 2022 and 2023. The local canopy at this elevation can be categorized as a deciduous hardwood forest, dominated by sugar maple, yellow birch and American beech trees. These trees are known to be strong monoterpene emitters with isoprene emissions depending on leaf age and heat stress (Karl et al., 2004; Jaakkola et al., 2024). In this study, we report only isoprene, a short-lived, highly reactive BVOC that is also prone to wall loss in canisters, making it a challenging compound to measure accurately. Grab samples were analyzed using the NSF NCAR Trace Organic Gas Analyzer (TOGA), a gas chromatograph coupled with a Time-of-Flight mass spectrometer (GC-MS), following the method described by Apel et al. (2003). At the WFM summit, trace gases are measured continuously throughout the year, including chemical species such as ozone (O3) and carbon monoxide (CO). Black carbon (BC) is measured by Aethalometer. Further details about the gas-phase dataset can be found in Brandt et al. (2016).
2.3 Sample handling procedures
2.3.1 Assessing short-term stability of LMWOA for a cloud water sample
As an initial test of the stability of organic acids within cloud water following collection prior to completing the chemical analyses, a set of tests was conducted by obtaining a filtered and unfiltered aliquot for a cloud water sample collected on 20 August 2019. The pH for this cloud water sample was 4.9. Both aliquots were split into two additional aliquots that were aged for four days before chemical analysis. The resulting measurements showed <10 % difference in pH, conductivity, , DOC, and concentrations after four days of aging and no significant difference in oxalate or formate between the filtered versus unfiltered aliquots (Table S4). However, the aged filtered sample exhibited 45 % higher acetate concentration than the aged unfiltered sample. This difference was not due to loss of acetate in the unfiltered sample, since the aged unfiltered sample had 11 % higher acetate concentrations than the fresh unfiltered sample. This puzzling result suggests possible microbial contamination being introduced during or after filtration with an Acetobacter species (He et al., 2022). While filtration is meant to prevent microbial degradation, utmost care must be taken to ensure that the additional sample handling does not introduce new microbes to the sample. However, this result could also reflect analytical artifacts rather than true acetate concentration, given such low absolute concentration and potential coelution of acetate with other organic acids, which we were unable to verify later.
2.3.2 Assessing short-term stability of LMWOA for cloud water mimic solution
To more carefully assess the impact of sample handling on measured organic acid concentrations, a field calibration with a known solution of organic acid concentrations is needed. An initial spray test with sodium oxalate test solution was conducted in 2018, but the results were compromised by the presence of wildfire smoke during the spray test at the WFM summit and failure to retain an aliquot of the original prepared solution for comparison. A carefully planned spray test of the cloud water collector with freshly prepared aqueous solution was conducted at the summit of WFM on a clean, cloud-free day on 22 September 2024 to systematically evaluate potential contamination, volatile loss and/or degradation of LMWOA during and shortly after sample collection. Following the bottle washing procedure for cloud water samples, the HDPE bottles used in the test were washed with hot water, soaked with ultrapure water for 48 h and subsequently confirmed free from contamination by IC analysis. A standard mixture solution (oxalate + formate + acetate + sulfate) was prepared with concentrations of 0.25 mg L−1 for each LMWOA and 0.4 mg L−1 for sulfate. The concentration of each analyte of the solution was verified in the lab by IC in the early morning prior to the spray test. All the measured concentrations of organics showed >95 % recovery of their original concentrations, while sulfate corresponded to 94.9 % recovery. The organic anions were added in their fully deprotonated forms by using disodium oxalate, sodium formate and sodium acetate certified Ion Chromatography stock solutions. Sulfate standard was prepared from ammonium sulfate in the lab and was added to the mixture to mimic cloud sample composition, since it has been a stable anion found in the samples over the years. The measured pH of the spray test solution was 6.05. At this pH, formate (pKa 3.75), acetate (pKa 4.76), and oxalate (pKa1 1.27, pKa2 4.27) remain almost entirely in their deprotonated anionic forms, which doesn’t favor volatilization of these LMWOA. It should be noted that the annual median pH of real cloud water samples ranges from pH 4.5 to 6.5 at WFM.
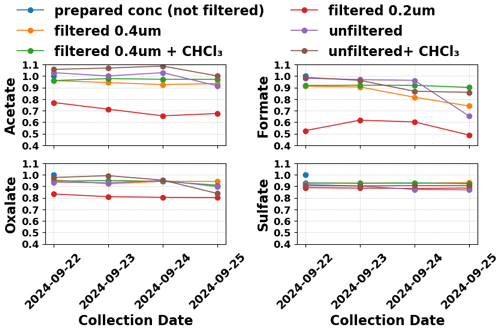
Figure 1Trends of measured analyte concentrations normalized by measured analyte concentrations of a prepared aqueous cloud water mimic solution, after spraying, collecting and aging over three days for the spray test.
Prior to the spray test, the cloud water collector was rinsed with ultrapure water for one minute, with rinse water going to waste. Then a rinse sample was collected by rinsing the entire system, including the in-line filter holder, with an additional ∼4 L of ultrapure water. Then approximately 2 L of the prepared test solution was sprayed on the collector and collected from the accumulator, which was labeled as “unfiltered” aliquot. The aliquot was split carefully into (five) 1 L ISCO bottles. In one of those, ∼50 µL chloroform (CHCl3) was added to assess microbial degradation. Two of the unfiltered aliquots were manually filtered separately using two different sizes of filters (0.2 and 0.4 µm), driven by the peristaltic pump. The 0.4 µm filtered aliquot was split into two ISCO bottles, and one was injected with 50 µL CHCl3. Finally, all five categories of aliquots were left in the accumulator fridge in labeled ISCO bottles to be collected and analyzed in the lab for the next three days to mimic the real cloud water sample collection process. The types of aliquots were (1) unfiltered, (2) unfiltered with added CHCl3, (3) filtered with 0.2 µm filter, (4) filtered with 0.4 µm filter, and (5) filtered with 0.4 µm filter with added CHCl3. A set of five samples was poured into the HDPE bottles on the same day, and the rest of the aliquots were left capped in five separate ISCO bottles for 12 h in the carousel to treat as if they were in the enclosed accumulator. The next day, again a set of five samples was collected, and ISCO bottles were left uncapped for the next two days. Samples from days 2 and 3 were kept frozen after collection for a few days before analysis to mimic the handling of cloud water samples.
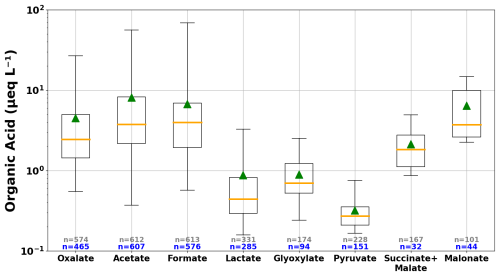
Figure 2Box and whisker plots of organic acid concentrations in cloud water samples collected at WFM in 2018–2024, for all measurements above detection limit. The number of samples in each year is reported as n (in grey font) and n (in blue font) for the total number of samples analyzed and the number of samples above detection limit, respectively. The orange lines mark median concentrations, green triangles mark mean concentrations, boxes indicate 25th and 75th percentiles, and whiskers extend from the 1st to 99th percentiles. Lactate measurements were unavailable in 2023–2024; glyoxylate and pyruvate measurements were unavailable in 2019 and 2023–2024; and succinate + malate concentrations were measured in 2018–2019 and malonate in 2018 only.
No significant organic contamination was detected in ultrapure water or rinse sample, though trace chloride was present. DOC measurement of the rinse sample was 150 ppb as compared to 750 ppb DOC for the spray solution and compared to the range of annual median cloud water DOC concentrations of 3600–6200 ppb from 2018–2024 (Fig. S6). A significant loss of formic and acetic acid was observed in the 0.2 µm filtered samples (Fig. 1), likely due to volatilization during the unexpectedly prolonged (>1 h) filtration process, during which time the sample was exposed to ∼20 °C lab air, in contrast to the normal cloud water sampling process. Filtered samples exhibited more stable organic acid concentrations. Unfiltered samples showed a decline in all LMWOA concentrations on the 3rd day. As a sanity check, replicates were run for the unfiltered and filtered (0.4 µm) samples, with results falling within ±5 %.
The observed decrease in LMWOA concentrations for unfiltered samples supports loss of organic acids by microbial activity in the unfiltered sample, especially for formate, with ∼35 % loss three days after collection. Although no microbes were intentionally introduced, later analysis done by Lombardo et al. (2025) confirmed the presence of microbes in 0.2 µm filtered samples using 16S DNA sequencing. Since the 0.4 µm filtered samples with CHCl3 added did not exhibit loss of formate by the 3rd day, microbial activity is likely responsible for the observed organic acid loss, rather than volatilization, for the 0.4 µm filtered samples. However, the pH of the individual sample determines how much volatilization of LMWOA may occur. Refrigeration at 1.1 ± 0.78 (1σ) °C within the accumulator fridge also helps reduce the rate of LMWOA volatilization.
Overall, the spray test confirms that short-term storage in the accumulator fridge (up to 3 d) is minimized if microbial degradation is suppressed by filtration and/or chloroform addition, with normal precautions to prevent contamination or loss by volatilization. The 0.4 µm filtered test sample experienced up to ∼16 % loss for formate, but no significant change in acetate or oxalate concentrations. Nearly all cloud water samples presented in this paper were collected using 0.4 µm filtration. The 0.2 µm filtered samples that began in 2024 are expected to experience less microbial degradation than the 0.4 µm filtered samples, and no volatile losses when the sampling system is intact, as during normal cloud water collection with automated in-line filtration.
3.1 Statistical summary of LMWOA 2018–2024
Formate and acetate are the most abundant monocarboxylic acids measured in WFM cloud water, with median concentrations of 3.95 and 3.75 µeq L−1 respectively, shown in Fig. 2. Oxalate and malonate are the next two abundant dicarboxylic acids, with median concentrations of 2.17 and 3.74 µeq L−1 respectively. Malonate was only measured in 2018 and may have a high bias, since co-elution with an unidentified peak precluded reliable measurements in the later years of the study. The rest of the organic acids measured had median concentrations <1 µeq L−1 and were frequently below detection limits, indicating their limited contributions to chemical properties in cloud water such as ion balance and acidity. For the remainder of the paper, the discussion of LMWOA focuses on formate (HCOO−), acetate (CH3COO−), and oxalate (), given that these three organic acids were measured for the entire seven-year period of this study.

Figure 3Relationship between measured DOC and oxalate concentrations in cloud water, colored by the average measured ozone (O3) mixing ratios during in-cloud periods for each sample, with marker shape representing the year that samples were collected. The slopes and correlation coefficients correspond to linear fits for time periods when O3<50 ppbv (blue) and O3>50 ppbv (red), with shaded areas representing the 95 % confidence band of the linear fits. Black “+” markers indicate time periods when in-cloud O3 mixing ratios could not be well characterized, either due to missing O3 or missing cloud liquid water content data.
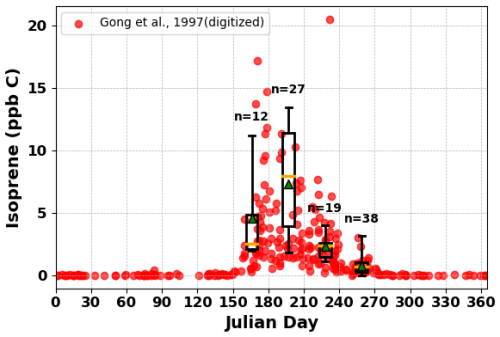
Figure 4Measured isoprene concentrations as sampled from the base of WFM throughout the year in 1994–1995 (red points, digitized from Gong and Demerjian, 1997) and throughout the summers of 2022 and 2023 (monthly box plots, with whiskers extending from 1st to 99th percentiles). The values of n correspond to the number of samples included in each box for each summer month (June, July, August, September) of the current study.
Monthly differences in LMWOA concentrations were observed for the three species of primary focus, as highlighted in Fig. S4. Formate, acetate, and oxalate concentrations exhibited the highest concentrations in the growing season (June and July) compared to August and September; here growing season corresponds to early summer, when canopy growth and leaf expansion are most active. Oxalate concentrations exhibited greater variability in July, when wildfire smoke events occurred more frequently, particularly in 2021 and 2023. These smoke events could provide additional precursors such as glyoxal, pyruvate, methylglyoxal, other LMWOA, further accelerating its dominant production pathway via in-cloud secondary processes, and aqueous phase reactions involving OH radical, as well as iron-catalyzed oxidation (Zuo and Zhan, 2005; Carlton et al., 2007; Kundu et al., 2010). The same wildfire smoke influence further explains both oxalate and DOC mean concentrations peaking in July. Also, a major fraction of in-cloud secondary formation of oxalate is known to originate from isoprene-derived compounds globally (Myriokefalitakis et al., 2011), which peaked in July at the site (Fig. 4). This contrasts with formate and acetate, which peaked in June, likely due to direct photochemical oxidation of a broad mix of vegetation and soil emissions under maximum sunlight exposure, with possibly a higher morning OH from HONO photolysis and more persistent nighttime NO3 radicals that could accelerate the primary production of those in the aqueous phase (Nguyen et al., 2015; Alwe et al., 2019). The decline in all LMWOA concentrations in September suggests reduced emission of precursors from the surrounding vegetation in the fall, which was also observed in isoprene measurements as shown in Fig. 4.
Oxalate generally accounts for ∼1 % of cloud water DOC concentrations. The measured oxalate to DOC ratio also exhibits an increase with measured ozone (O3) mixing ratios, especially apparent in cloud water samples collected in 2018. However, the relationship to O3 is less clear in subsequent years. Overall, for cloud water samples collected 2018–2024, the oxalate : DOC ratio increases only slightly from (1.0 ± 0.084) % to (1.4 ± 0.27) % for time periods with <50 and >50 ppbv O3, respectively, as shown in Fig. 3. Higher O3 levels can enhance secondary oxalate formation from its gas phase precursors such as glyoxal, methylglyoxal and pyruvic acid (Kawamura and Bikkina, 2016) via BVOC oxidation pathways, as observed in an aerosol study from a forest at Mt. Fuji (Mochizuki et al., 2017). However, higher DOC concentrations may alter oxidant mixing ratios, and extreme smoke events (typically associated with high DOC concentrations) can substantially reduce actinic flux (Park et al., 2018; Ervens, 2022), which can complicate the relationship between oxalate, DOC and O3, likely accounting for some of the variability observed in Fig. 3.
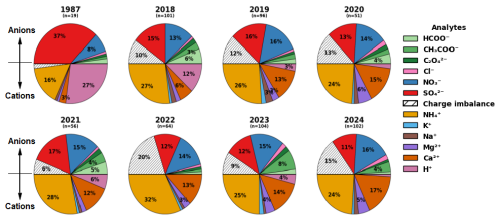
Figure 5Annual mean fractions of ion concentrations (in eq L−1) of measured cations and anions in cloud water for 1987 (Khwaja et al., 1995) and 2018–2024 (this study). Only the subset of samples for which measurements of all listed analytes were obtained is included in the mean (including analytes with measured concentrations below MDL), and the number of samples included in the mean is reported as n for each year. Note that the 1987 data are from hourly samples from three summer cloud events, while the 2018–2024 dataset represents 12-hourly summertime measurements obtained continuously from June through September in each year, except for 2021, which corresponds to samples collected June through August.
Figure 4 shows a comparison of isoprene concentrations measured at WFM in 1994–1995, reported in Gong and Demerjian (1997), overlaid with our 2022–2023 data at WFM shown in a box-whisker plot. The 2022–2023 data have a higher mean in July (∼7.31 ppb C) compared to Gong and Demerjian (1997) (∼3.65 ppb C), possibly due to differences in sampling frequency, but also due to the inherent variability of isoprene due to its high reactivity. However, both datasets follow a similar seasonal trend, peaking in July and declining in fall, which aligns with the monthly trends of all three major LMWOA and DOC, suggesting that isoprene emissions play a key role in the production of LMWOA.
3.2 Contribution of LMWOA to Ion Balance
Ion fractions are averaged by year in Fig. 5. Major changes in cloud water ion fractions between 1987 (Khwaja et al., 1995) and recent years include a decrease in sulfate ion and increases in ammonium and calcium. The three most abundant LMWOA account for 6 %–22 % of anions and 3 %–9 % of DOC on an annual basis (Figs. 5 and S3). On average, 14 %–40 % of anions remain unaccounted for, which could include other organic anions such as organosulfates, organonitrates, malonate, succinate, glutarate and other dicarboxylic acids, as well as other potential anions like halides and bicarbonate. High molecular weight multifunctional organics like Humic-Like Substances (HULIS), produced through aqueous phase reactions of small aldehydes with ammonium and amines during cloud processing and oligomerization of water-soluble organics during cloud processing of wildfire smoke (Salma et al., 2008; Hawkins et al., 2016; Laskin et al., 2025), may also contribute to both the unidentified DOC species and ion imbalance. Assessing ion balance for all samples (2018–2024), the three major LMWOA increase the slope for measured anion versus cation concentrations from 0.61 to 0.72 over the seven years (Fig. S5).
3.3 LMWOA and “Surplus Ammonium” in Cloud water
In a growing fraction of cloud water samples, ammonium concentrations exceed the combined concentrations of sulfate and nitrate (i.e., ). This shift is described by Lawrence et al. (2023) as a “New Chemical Regime” where the surplus is no longer fully neutralized by strong inorganic acids, as also discussed by Pye et al. (2020). Under this regime, we observe a strong positive correlation between surplus and the sum of three major LMWOA such as formate, acetate, and oxalate (Fig. 6). In contrast, samples with show weak or no correlation between these species. This suggests that when surplus is available, it interacts more with deprotonated organic acids, forming ammonium organic salts, especially in DOC-rich samples.
High surplus concentrations associated with high LMWOA concentrations occurred during a heat wave in 2018 (Fig. 6), which correlated with both high ozone mixing ratios and high BVOC concentrations, likely resulting in enhanced secondary formation of LMWOA (Lawrence et al., 2024). However, for DOC-rich samples collected in 2023 and 2024, the relationship between surplus and LMWOA concentrations is much less clear, perhaps in-part due to differences in NH3 availability and nucleophilic addition reactions between NH3 and organic acids, which can produce diverse nitrogen-containing organic compounds (which are not included in our dataset) but may act as a sink for both and some organic acids (Shu et al., 2025). The high DOC events from 2021 and 2023 are associated with two extreme wildfire smoke events, which likely follow chemically distinct aging pathways as compared to the 2018 pollution event modeled by Lawrence et al. (2024). We discuss and contrast the chemical and meteorological conditions encountered during these two wildfire smoke events in Sect. 3.5 of this paper.
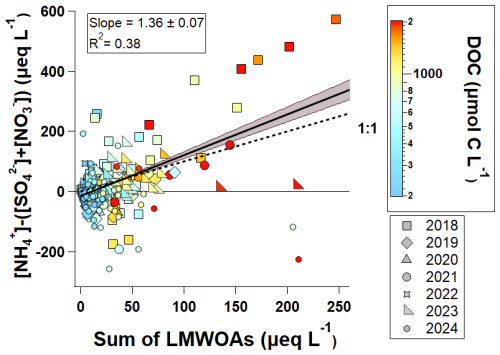
Figure 6Relationship between surplus and sum of three major LMWOA measured in cloud water collected in 2018–2024, colored by the measured DOC concentrations for each sample; the shaded area represents the 95 % confidence band and the slope corresponds to the linear fit. Again, the LMWOA values reported here represent the measured concentrations, without accounting for dissociation state.
3.4 Contribution of LMWOAs to cloud droplet acidity
The significant contribution of organic acids to ion balance suggests that organic acids can also be important for cloud droplet acidity. Lawrence et al. (2023) used a “top down” vs. “bottom up” framework to estimate cloud droplet acidity based on the measured inorganic ions. By comparing pH from these two different calculations, a Missing Acid Fraction (MAF) was inferred, which was shown to be rapidly growing at WFM since ∼2009, with more than half of the inferred cloud droplet acidity no longer explained by the traditional inorganic ions , and when that study concluded in 2021. They proposed that unmeasured LMWOA could be a dominant species reshaping the site’s chemical regime. The updated dataset presented here shows that the difference between pHTD (blue) and pHBU (orange) has continued to grow (Fig. 7), resulting in a further increase in MAF since 2021 (Fig. 8) and approaching MAF=1.0 in 2022–2024, with nearly all of the inferred cloud droplet acidity unaccounted for by the measured inorganic ions. Note that pHTD varied very little during the entire seven-year study, with annual median values averaging ∼4.4 ± 0.09 (1σ).

Figure 7Annual median cloud water pH (2018–2024) using four different approaches: (1) measured bulk cloud water pH (black), (2) “top down” pH (pHTD) derived from measured bulk pH and measured Ca2+ and Mg2+ concentrations (blue) with error bars representing one standard deviation in pHTD for each year, (3) “bottom up” pH (pHBU) derived from measured concentrations of , and as originally defined by Lawrence et al. (2023) (orange), and (4) a new “bottom up” pH (pHBU*) that also includes the dissociated LMWOA concentrations calculated from their measured concentrations: formate, acetate and oxalate (red). Note that pH measurements have 5 % precision and 98 %–102 % accuracy, as detailed in the Supplement.

Figure 8Annual median fraction of samples with surplus (i.e. in red on the left axis. Annual median Missing Acid Fraction (MAF) on right axis, calculated in two different ways: (1) MAF as originally defined by Lawrence et al. (2023), including only measured inorganic ions (black solid line), with negative ([H+]BU values set to zero), and (2) newly defined MAF* including dissociated LMWOA (black dotted line), with negative ([H+]BU* values set to zero). Also plotted is a linear fit to the annual median MAF* (blue dashed line). The blue shaded box plot shows the variability in MAF* in each year, with the upper edges of the boxes representing the 75th percentile and the lower edges of the boxes representing the 25th percentile. The MAF* error bars show the estimated uncertainty in MAF*, with the upper error bar including the addition of measured K+ concentrations in HBU* calculation and the lower error bar including the measured LMWOA concentrations (formate, acetate and oxalate) assuming full deprotonation regardless of pH.
Adding the new measurements of LMWOA to the “bottom up” calculation for estimated cloud droplet pH decreases pHBU* by 0.1–0.45 units and explains 5 %–36 % of the cloud droplet acidity on an annual median basis. These findings are supported by previous studies where formic + acetic acids supplied up to 60 % of droplet acidity in remote and mountain sites (Keene et al., 1983; Galloway et al., 1982; Keene and Galloway, 1986; Andreae et al., 1988; Pye et al., 2020). The contribution of LMWOA to droplet acidity was greatest in 2018, 2021 and 2023, as exhibited by a greater difference between MAF and MAF* in Fig. 8. The latter two of these years were heavily influenced by high primary LMWOA emissions from wildfire smoke events (Permar et al., 2023), with LMWOA contributing 30 %–33 % to cloud droplet acidity on an annual median basis in 2021 and 2023. The meteorological and chemical properties associated with wildfire smoke events in these two years are contrasted in Sect. 3.5 of this paper. However, the even stronger impact of LMWOA on cloud droplet acidity in 2018 likely resulted from different precursors and dominant reaction pathways than in 2021 and 2023, since the 2018 pollution event modeled by Lawrence et al. (2024) was associated with high temperatures, high O3 and high BVOC concentrations, with little influence from wildfire smoke.
The linear fit to annual median MAF* (which includes dissociated LMWOA) exhibits an upward trend with a 7.6 % increase per year and r2=0.59 over the seven years of measurements (Fig. 8). The upper error bars represent MAF* with measured K+ concentrations added to the HBU* calculation to account for potential internal mixing of K+ in the fine aerosol mode. While K+ may be found in both the fine and coarse aerosol particles (VandenBoer et al., 2011), Adachi et al. (2022) showed that, in wildfire smoke, K+ is often internally mixed with fine organic aerosol. Figure 8 shows that the uncertainty in MAF* due to K+ mixing state is typically small compared to other sources of uncertainty. The lower error bar for MAF* represents the maximum possible contribution of measured LMWOA, assuming complete dissociation of these acids irrespective of pH. In the years 2021 and 2023 when wildfire events were more frequent, higher uncertainties are observed for the contribution of LMWOA to MAF* (Fig. 8). Another source of uncertainty in the missing acid fraction calculation arises from the assumption that is entirely in fine mode aerosol, whereas in reality some fraction may exist in coarse mode aerosol (Lee et al., 2008). Note that if were alternatively found entirely within the coarse mode, the MAF* calculation would likely be minimally affected, since and since both HTD and HBU* would be overestimated by the same degree, exactly equal to the measured bulk concentrations.
3.5 Influence of Smoke on Cloud Water Composition
Wildfire smoke events are expected to grow more frequent with climate change (Burke et al., 2023). Smoke plumes are known to be highly enriched in organic matter (Liu and Peng, 2019) and the associated particles can be acidic (Smolyakov et al., 2014; Behera et al., 2015). While NH3 emissions are often thought to be associated with agriculture (e.g., fertilizers and animal waste), biomass burning plumes can also be enriched in NH3 (Bray et al., 2018). Thus, smoke-influenced cloud events present interesting case studies for more in-depth analysis of LMWOA, surplus and MAF under high DOC loadings. Summertime smoke plumes over regions such as Saskatchewan, Manitoba, Ontario, and Quebec are dominated by emissions from wildland Boreal fires (Brey et al., 2018), frequently transported to WFM. In this section, two severe wildfire smoke episodes are compared to assess how smoke plume aging shapes the chemical composition of cloud water samples. Both of these smoke events were associated with Boreal forest fires in Canada, but the fires were located at different distances upwind from WFM, thereby impacting the transport time and transport path of the smoke plume to our site.
The first smoke event, which intercepted WFM on 19–22 July 2021, was detected across NY State with ground-based LIDAR measurements (Shrestha et al., 2022). Zheng et al. (2023a) reported record-breaking Boreal wildland forest fire emissions in 2021, with Manitoba, Saskatchewan, British Columbia, and Ontario provinces witnessing their largest total area burned in July 2021 (Jain et al., 2024b), intensified by a Pacific Northwest heat dome. HYSPLIT back trajectories, launched at 12:00 UTC on 20 July 2021 from the WFM summit (Lawrence et al., 2023) traced that plume back to south-central Canada with at least five days transport time from the source to the measurement site. Hence, we categorize this event as an “Aged Smoke” plume, and three cloud water samples were collected during this episode. We report LMWOA data from two of those samples since not enough sample volume was collected in the first sample to conduct all chemical analyses, since clouds were intercepted only at the very end of the 12 h sampling period (as indicated in yellow shading as the daytime period on 19 July in the LWC panel of Fig. 9). Nevertheless, enough sample volume was collected to conduct all other measurements.
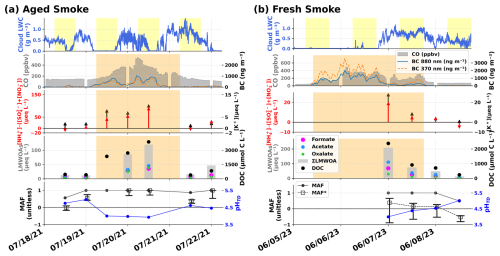
Figure 9Time series of cloud water compositions during smoke-influenced periods at Whiteface Mountain in (a) 18–22 July 2021 and (b) 5–8 June 2023. From top to bottom: (1) cloud liquid water content (LWC, g m−3); (2) carbon monoxide (CO, ppbv) and black carbon (BC, ng m−3); (3) surplus ammonium (, µeq L−1) and potassium ([K+], µeq L−1); (4) Measured LMWOA concentrations (oxalate, acetate, formate, and ∑LMWOA in µeq L−1) along with DOC, µm C L−1); and (5) MAF (inorganics only) and MAF* (inorganics + LMWOA) with estimated cloud droplet pHTD. The upper error bar of MAF* includes the addition of measured K+ concentrations in HBU* calculation and the lower error bar includes the measured LMWOA concentrations assuming full deprotonation regardless of pH. Daylight hours (06:00–18:00 EST) are indicated with yellow shading in the top graph and smoky periods are indicated by brown shading in the bottom three graphs, as determined from the measured K+, CO and BC concentrations.
The second smoke event originated from lightning-caused wildfires from south-central Quebec (Jain et al., 2024a). These fires produced a colossal smoke plume that caused severe air pollution and record-breaking daily mean PM2.5 concentration in New York City (NYC) on 7 June, making it the city's worst air quality event in the last 50 years (Wang et al., 2024; Cooper et al., 2024). The smoke plume arrived in New York City (Queens) in a mean of 47.5 h per the HYSPLIT forward trajectory, severely affecting the air quality of NYC from 6–9 June 2023 (Joo et al., 2024). Since WFM lies upwind from NYC, and showed elevated carbon monoxide (CO) and black carbon (BC) levels roughly 24 h before the plume reached the city, we estimate this smoke was about one day old, significantly fresher than the first event. Hence, it has been categorized as a “Fresh Smoke” event influencing two cloud water samples collected during that period. The woodsmoke contribution (obtained by subtracting the 370 nm absorption channel from the 880 nm absorption channel of the aethelometer used to measure BC, Allen et al., 2004) was markedly higher for this event than the “Aged Smoke” event. Note that, since BC can be scavenged in clouds, and cloud droplets >10 µm are not sampled by the aerosol inlet (Lance et al., 2020), the BC measurements during in-cloud periods should be treated as a lower bound of the actual BC mass loading since only interstitial aerosols and smaller cloud droplets containing BC are sampled by this instrument while in-cloud. Interestingly, the second sample from this “Fresh Smoke” event appeared to have higher BC and woodsmoke concentrations than the first sample, potentially indicating less efficient scavenging of smoke particles by cloud droplets, despite the high LWC, possibly due to a change in the smoke plume mixing-state and/or particle size distribution.
Elevated concentrations of biomass burning tracers, including CO, BC, DOC and K+ coincide with both elevated surplus and LMWOA concentrations for these two smoke events, reinforcing the previously discussed chemical interactions between surplus and LMWOA, but specifically for wildfire smoke plumes here. However, there are some marked differences between these two episodes. The “Aged Smoke” samples exhibited substantially higher surplus than the “Fresh Smoke,” despite cloud droplets from both events exhibiting the same high acidity levels (pHTD∼3.9) and liquid water content (LWC), which both favor NH3 partitioning to the aqueous phase. This difference may be explained by the different transport paths and aging times of the smoke plumes. The “Aged smoke” plume was transported across the agriculturally intensive Upper Midwest (Wisconsin), where it appears to have picked up more NH3, resulting in surplus concentrations nearly as high as the measured LMWOA concentrations. The “Fresh Smoke” plume passed over northern NY and Montreal, where there is much less agricultural activity, likely leading to the much lower surplus concentrations observed in those samples, accounting for only about 10 % of the measured LMWOA concentrations. Samples from both events contained elevated levels of LMWOA, with “Fresh Smoke” samples richer in monocarboxylic acids (formate, acetate), which have been shown previously to be directly emitted by wildland fires (Permar et al., 2021, 2023).
The “Aged Smoke” samples exhibited lower LMWOA concentrations and a reduced LMWOA contribution to DOC compared to the “Fresh Smoke” samples. We hypothesize that for “Aged Smoke” samples, a major fraction of the freshly emitted monocarboxylic acids had undergone aqueous phase transformation as the plume aged, resulting in lower LMWOA concentrations several days later when the samples were intercepted at WFM. Conversely, DOC concentrations in the “Aged Smoke” samples were significantly higher than the “Fresh Smoke” samples. A simultaneous decrease in LMWOA and increase in DOC may suggest that oligomerization or accretion reactions dominate over fragmentation reactions during atmospheric aging (Barsanti and Pankow, 2004; Jimenez et al., 2009), boosting DOC during transport and potentially forming CHON compounds (Jin et al., 2026), as also observed in a previous study on Boreal wildfire smoke plume aging (Ditto et al., 2021). Functionalization reactions can further enhance DOC, since oxidation of insoluble BC and Brown Carbon (BrC, Sullivan et al., 2022) can also increase DOC loading upon aging (Hems et al., 2020; Gilardoni et al., 2016), consistent with both the greater DOC and lower BC and woodsmoke signals observed in the “Aged Smoke” samples. CO mixing ratios were similar during both events, peaking at ∼500 ppbv, but generally in the 200–400 ppbv range.
When including LMWOA in the estimated cloud droplet pH calculation, 70 %–90 % of the Missing Acid Fraction (MAF*) was resolved in the “Fresh Smoke” samples (Fig. 9b, bottom panel), illustrating the importance of these measured compounds to cloud droplet acidity. However, for the “Aged Smoke” samples, the estimated cloud droplet acidity remained entirely unaccounted for, even after including LMWOA in the MAF* calculation (Fig. 9a, bottom panel). This difference indicates that cloud droplet acidity for the “Fresh Smoke” event is primarily driven by LMWOA (formate, acetate, oxalate), whereas cloud droplet acidity for the “Aged Smoke” event is dominated by other unmeasured anions, likely also organic-rich, including higher molecular weight, possibly multifunctional organic acids. Since these were smoke-influenced samples, the potential effect of K+ on pHBU was tested, with impact on MAF* represented by the upper error bars shown in bottom panels of Fig. 9. Again, we also evaluated the maximum possible contribution of LMWOA concentrations to pHBU by assuming full deprotonation regardless of pHTD, with impact on MAF* represented by the lower error bars shown in Fig. 9. It has been reported before that K+ participates in neutralizing cloud water acidity, potentially forming potassium organic salts (Jing et al., 2017).
Cloud droplet acidity was high for both of the highlighted smoke events, with pHTD<4.0 in most of these cloud water samples, matching median bulk cloud water pH from the 1990s prior to full implementation of the CAA amendments (Lawrence et al., 2023), as compared to pHTD values ≳4.5 in cloud water samples collected before and after these two smoke events. With the formation of higher molecular weight organic acids during the aging process, any new production of carboxylic acid functional groups could further lower pH. However, for the “Aged Smoke” event, cloud droplets acquired additional through gas phase partitioning of NH3, neutralizing any added protons (Schwab et al., 2016; Shah et al., 2020; Tilgner et al., 2021; Driessen and Murphy, 2025). This external buffering by ammonia uptake explains the higher surplus in “Aged Smoke” samples at the same cloud droplet pH and LWC level as “Fresh Smoke” samples, a mechanism that is distinctly different in aerosols and clouds, as discussed by Zheng et al. (2023b).
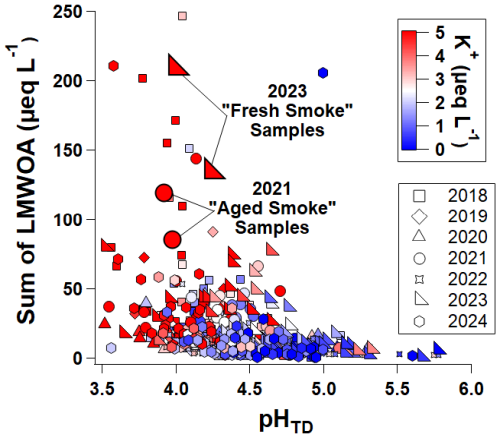
Figure 10Measured LMWOA concentrations versus estimated cloud droplet pH, colored by the measured potassium ion concentrations, with marker shape indicating the year that cloud water samples were collected from WFM. The two extreme smoke events highlighted in Fig. 9 are emphasized here with larger markers, to visualize their relationship within the broader context of the seven-year dataset.
We further evaluate smoke-influence on measured LMWOA concentrations for the full seven-year dataset, using measured K+ concentrations >2.5 µeq L−1 as a proxy for smoke-influence, consistent with mean K+ concentrations for cloud water samples characterized as high probability of smoke-influence by Lee et al. (2022). Figure 10 shows that LMWOA concentrations >50 µeq L−1 generally occur at pHTD values <4.7, coinciding with high K+ concentrations. This suggests that smoke-influenced cloud droplets are moderately acidic, often likely in part due to their high organic acid concentrations. A similar relationship between DOC and pHTD, fit to an exponential decay function, was reported by Lawrence et al. (2023), with smoke-influenced samples from 2021 exhibiting the highest DOC values observed throughout that previous WFM study (note that cloud water samples from 2022–2024 were not included in that study). The review article by Pye et al. (2020) also characterized biomass burning aerosol as drivers of acidity in cloud droplets. A previous analysis of cloud water samples collected from WFM in 2014 (Cook et al., 2017) showed an increase in O:C ratio as the bulk cloud water pH decreased, with two out of three of the smoke-influenced samples exhibiting the lowest bulk pH values, even more acidic than the urban-influenced cloud water samples selected in that study. They further reported that smoke-influenced samples contained more oxidized and chemically complex dissolved organic matter, including slightly higher average O:C ratios and double bond equivalent (DBE) compared to non-smoke-influenced samples (e.g., biogenic, urban, or mixed source). This previous analysis, which focused on higher molecular weight organic compounds, is consistent with the observed increase in LMWOA with cloud droplet acidity in the current study, suggesting that aqueous phase oxidation of both low molecular weight and high molecular weight compounds occurs simultaneously under acidic conditions.
While measured K+ concentrations in cloud water have been linked to smoke-influence, the available cloud water dataset cannot be used to directly ascertain the age of smoke plumes. The two extreme smoke events highlighted in this study suggest that atmospheric aging of smoke during transport over 1–5+ d can substantially alter the chemical properties of the smoke-influenced cloud water, consistent with the findings of Joo et al. (2024), who found that aged smoke plumes were at risk of being misclassified due to the chemical transformations occurring during the aging process. Future work utilizing comprehensive backtrajectory analysis, in conjunction with satellite observations and potentially other in-situ chemical markers like NOx and NOy, is needed for robust assessment of smoke plume age on LMWOA concentrations and cloud droplet pH.
This study presents a comprehensive seven-year summertime dataset of three major Low Molecular Weight Organic Acids (LMWOA, i.e., formate, acetate and oxalate) in cloud water collected via semi-automated sampling from atop Whiteface Mountain (WFM) 2018–2024. While multiple labs and methods were utilized to complete these long-term measurements, the analytical methods used in the study are appropriate, transparent and reproducible. Based on a systematic spray test of the cloud collection equipment at the WFM summit with prepared aqueous solution of known sulfate and organic acid concentrations, and repeated analyses after long-term freezer storage, we show that frequent rinses, in-line filtration, refrigeration and timely analysis are crucial for preservation of LMWOA, to minimize both volatilization and microbial degradation prior to offline chemical analyses.
The present study offers insights into the seasonal variability and sources of LMWOA, as well as assessing relationships between LMWOA, Dissolved Organic Carbon (DOC), surplus ammonium and acidity. Measured formate and acetate concentrations exhibited seasonal changes consistent with local emissions of Biogenic Volatile Organic Compounds (BVOC). The robust relationship between oxalate and DOC, with slightly higher slope under high oxidant loadings (associated with elevated ozone mixing ratios), is consistent with in-cloud secondary production of oxalate from its precursors, e.g., volatile dicarbonyls like glyoxal and methylglyoxal (Ervens et al., 2011; Lim et al., 2013). In our dataset, the oxalate-to-sulfate ratio (slope=0.073, Fig. S8) is higher than the global aerosol median oxalate-to-sulfate (0.0217) reported by Hilario et al. (2021), while still falling within the broader variability observed across worldwide aerosol campaigns, where the highest values (∼0.35) were associated with anthropogenically polluted air masses and sometimes influenced by coarse aerosol or smoke and lowest values (∼0.01) corresponded to cleaner, less polluted or weakly processed air masses.
The correlation between surplus ammonium and LMWOA reinforces their linkage in cloud chemistry. LMWOA can pair with (Sorooshian et al., 2006; Herrmann et al., 2015) after neutralizes sulfate () and nitrate (). The growing surplus of relative to and highlights that the role of organic acids in ammonium partitioning has changed, driven in-part by long-term reductions in SO4 concentrations due to dramatic reductions in SO2 emissions (Lawrence et al., 2023), while NH3 emissions have remained stable or may have even increased (Guo et al., 2017; Yu et al., 2018).
In spite of significant improvements to ion balance with inclusion of LMWOA in the present study, a significant ion imbalance remains. Furthermore, the missing acid fraction is shown to be growing on an annual median basis over this seven-year study, with the majority of cloud droplet acidity unaccounted for by the measured , , and LMWOA concentrations over the past several years.
The current study also highlights two extreme wildfire smoke events, both from Boreal wildland forest fires in Canada, but at two different locations upwind of WFM and in two different years. Comparison of the two events highlights the importance of transport path and aging on the wildfire smoke influence on cloud water chemical composition. Enrichment in LMWOA for the “Fresh Smoke” event drives cloud droplet acidity, whereas the acidity of cloud droplets in the “Aged Smoke” event remains entirely unaccounted for by the measured analytes (including LMWOA), suggesting that other unmeasured organic acids or other organic anions (Roberts et al., 2011) are contributing substantially to acidity, which would require additional chemical analyses to evaluate.
In this study, we present the first long-term dataset of LMWOA in North American cloud water, further documenting the well-known shift from inorganic to organic dominance in cloud water chemistry post Clean Air Act (CAA). The current study underscores the need to extend routine cloud water organic chemistry measurements to a broader suite of organic acids, as the LMWOA measured in this study is shown to play an important role in cloud droplet acidity, but a substantial and growing fraction of the cloud droplet acidity remains unaccounted for. FTIR spectroscopy would be a valuable addition to the analytical measurement suite since it can provide bulk total organic acid equivalents, which would complement the ion budget the most (Coury and Dillner, 2008).
Cloud water data can be found at http://atmoschem.asrc.cestm.albany.edu/~cloudwater/pub/Data.htm (Lance, 2023). Quality controlled air quality data for the WFM summit and base sites for the period 1989–2021 and associated data analysis code are available at https://doi.org/10.5281/zenodo.4394567 (May, 2020).
The supplement related to this article is available online at https://doi.org/10.5194/acp-26-3951-2026-supplement.
A. Tripathy analyzed the datasets and wrote the paper under the guidance of S. Lance and with contributions from all co-authors. P. Casson conducted the cloud water sampling with assistance from R. Brandt, S. Lance, C. E. Lawrence, N. Pittman, S. Lombardo and A. Tripathy. D. Kelting and E. Yerger conducted the cloud water chemical analyses including organic acids in 2018–2019. A. Tripathy conducted the organic acid analysis in 2020–2024, with training and assistance from H. Khwaja and M. Hussain. P. Snyder conducted chemical analyses of cloud water samples in 2020–2021, excluding organic acids. A. Tripathy conducted all other chemical analyses of cloud water samples 2022–2024 with assistance from S. Lombardo, R. Patel, L. Hammond and K. DeMarle. Ozone, carbon monoxide and black carbon datasets were acquired and quality controlled by R. Brandt, S. McKim and J. Schlemmer, with assistance from N. Pittman in 2022. VOC sampling was conducted by A. Tripathy and VOC measurements were conducted in collaboration with E. C. Apel, R. S. Hornbrook and A. J. Hills.
At least one of the (co-)authors is a member of the editorial board of Atmospheric Chemistry and Physics. The peer-review process was guided by an independent editor, and the authors also have no other competing interests to declare.
The New York State Energy Research and Development Authority (NYSERDA) has not reviewed the information contained herein, and the opinions expressed in this report do not necessarily reflect those of NYSERDA or the State of New York.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. The authors bear the ultimate responsibility for providing appropriate place names. Views expressed in the text are those of the authors and do not necessarily reflect the views of the publisher.
A. Tripathy and S. Lance gratefully acknowledge Michael Battaglia for his hands-on assistance and guidance with the Metrohm IC, as well as Jim Schwab and William May for providing the quality-controlled air quality dataset from Whiteface Mountain. The authors acknowledge the New York State (NYS) Department of Environmental Conservation (DEC) for the summit ozone measurements.
This research has been supported by the NSF Faculty Early Career Development (CAREER) grant (award number 1945563), NASA Future Investigators in NASA Earth and Space Science and Technology (FINESST) program (award number 80NSSC21K1633), NSF ACOM Ralph Cicerone Fellowship in Earth System Science (https://www2.acom.ucar.edu/ralph-cicerone-fellowship, last access: 18 March 2026) and New York State Energy Research and Development Authority (NYSERDA) contract 124461. Trace gas, particulate and meteorological measurements at Whiteface Mountain were supported by NYSERDA contract 48971. This material is based upon work supported by NSF National Center for Atmospheric Research (NCAR), which is a major facility sponsored by the U.S. National Science Foundation under Cooperative Agreement No. 1852977.
This paper was edited by John Liggio and reviewed by three anonymous referees.
Adachi, K., Dibb, J. E., Scheuer, E., Katich, J. M., Schwarz, J. P., Perring, A. E., Mediavilla, B., Guo, H., Campuzano-Jost, P., Jimenez, J. L., Crawford, J., Soja, A. J., Oshima, N., Kajino, M., Kinase, T., Kleinman, L., Sedlacek III, A. J., Yokelson, R. J., and Buseck, P. R.: Fine Ash-Bearing Particles as a Major Aerosol Component in Biomass Burning Smoke, J. Geophys. Res.-Atmos., 127, e2021JD035657, https://doi.org/10.1029/2021JD035657, 2022. a
Allen, G. A., Babich, P., and Poirot, R. L.: Evaluation of a new approach for real time assessment of wood smoke PM, in: Proceedings of the regional and global perspectives on haze: causes, consequences, and controversies, air and waste management association visibility specialty conference, 25–29, 2004. a
Altieri, K., Seitzinger, S., Carlton, A., Turpin, B., Klein, G., and Marshall, A.: Oligomers formed through in-cloud methylglyoxal reactions: Chemical composition, properties, and mechanisms investigated by ultra-high resolution FT-ICR mass spectrometry, Atmos. Environ., 42, 1476–1490, https://doi.org/10.1016/j.atmosenv.2007.11.015, 2008. a
Alwe, H. D., Millet, D. B., Chen, X., Raff, J. D., Payne, Z. C., and Fledderman, K.: Oxidation of Volatile Organic Compounds as the Major Source of Formic Acid in a Mixed Forest Canopy, Geophys. Res. Lett., 46, 2940–2948, https://doi.org/10.1029/2018GL081526, 2019. a
Amato, P., Demeer, F., Melaouhi, A., Fontanella, S., Martin-Biesse, A.-S., Sancelme, M., Laj, P., and Delort, A.-M.: A fate for organic acids, formaldehyde and methanol in cloud water: their biotransformation by micro-organisms, Atmos. Chem. Phys., 7, 4159–4169, https://doi.org/10.5194/acp-7-4159-2007, 2007. a
Andreae, M. O., Talbot, R. W., Andreae, T. W., and Harriss, R. C.: Formic and acetic acid over the central Amazon region, Brazil: 1. Dry season, J. Geophys. Res.-Atmos., 93, 1616–1624, https://doi.org/10.1029/JD093iD02p01616, 1988. a
Apel, E. C., Hills, A. J., Lueb, R., Zindel, S., Eisele, S., and Riemer, D. D.: A fast-GC/MS system to measure C2 to C4 carbonyls and methanol aboard aircraft, J. Geophys. Res.-Atmos., 108, 2002JD003199, https://doi.org/10.1029/2002JD003199, 2003. a
Baray, J.-L., Deguillaume, L., Colomb, A., Sellegri, K., Freney, E., Rose, C., Van Baelen, J., Pichon, J.-M., Picard, D., Fréville, P., Bouvier, L., Ribeiro, M., Amato, P., Banson, S., Bianco, A., Borbon, A., Bourcier, L., Bras, Y., Brigante, M., Cacault, P., Chauvigné, A., Charbouillot, T., Chaumerliac, N., Delort, A.-M., Delmotte, M., Dupuy, R., Farah, A., Febvre, G., Flossmann, A., Gourbeyre, C., Hervier, C., Hervo, M., Huret, N., Joly, M., Kazan, V., Lopez, M., Mailhot, G., Marinoni, A., Masson, O., Montoux, N., Parazols, M., Peyrin, F., Pointin, Y., Ramonet, M., Rocco, M., Sancelme, M., Sauvage, S., Schmidt, M., Tison, E., Vaïtilingom, M., Villani, P., Wang, M., Yver-Kwok, C., and Laj, P.: Cézeaux-Aulnat-Opme-Puy De Dôme: a multi-site for the long-term survey of the tropospheric composition and climate change, Atmos. Meas. Tech., 13, 3413–3445, https://doi.org/10.5194/amt-13-3413-2020, 2020. a
Barsanti, K. C. and Pankow, J. F.: Thermodynamics of the formation of atmospheric organic particulate matter by accretion reactions – Part 1: aldehydes and ketones, Atmos. Environ., 38, 4371–4382, https://doi.org/10.1016/j.atmosenv.2004.03.035, 2004. a
Barth, M. C., Rasch, P. J., Kiehl, J. T., Benkovitz, C. M., and Schwartz, S. E.: Sulfur chemistry in the National Center for Atmospheric Research Community Climate Model: Description, evaluation, features, and sensitivity to aqueous chemistry, J. Geophys. Res.-Atmos., 105, 1387–1415, https://doi.org/10.1029/1999JD900773, 2000. a
Barth, M. C., Ervens, B., Herrmann, H., Tilgner, A., McNeill, V. F., Tsui, W. G., Deguillaume, L., Chaumerliac, N., Carlton, A., and Lance, S. M.: Box Model Intercomparison of Cloud Chemistry, J. Geophys. Res.-Atmos., 126, e2021JD035486, https://doi.org/10.1029/2021JD035486, 2021. a, b
Behera, S. N., Cheng, J., and Balasubramanian, R.: In situ acidity and pH of size-fractionated aerosols during a recent smoke-haze episode in Southeast Asia, Environ. Geochem. Hlth., 37, 843–859, https://doi.org/10.1007/s10653-014-9660-1, 2015. a
Bianco, A., Riva, M., Baray, J.-L., Ribeiro, M., Chaumerliac, N., George, C., Bridoux, M., and Deguillaume, L.: Chemical Characterization of Cloudwater Collected at Puy de Dôme by FT-ICR MS Reveals the Presence of SOA Components, ACS Earth and Space Chemistry, 3, 2076–2087, https://doi.org/10.1021/acsearthspacechem.9b00153, 2019. a
Boone, E. J., Laskin, A., Laskin, J., Wirth, C., Shepson, P. B., Stirm, B. H., and Pratt, K. A.: Aqueous Processing of Atmospheric Organic Particles in Cloud Water Collected via Aircraft Sampling, Environ. Sci. Technol., 49, 8523–8530, https://doi.org/10.1021/acs.est.5b01639, 2015. a
Brandt, R. E., Schwab, J. J., Casson, P. W., Roychowdhury, U. K., Wolfe, D., Demerjian, K. L., Civerolo, K. L., Rattigan, O. V., and Felton, H. D.: Atmospheric Chemistry Measurements at Whiteface Mountain, NY: Ozone and Reactive Trace Gases, Aerosol Air Qual. Res., 16, 873–884, https://doi.org/10.4209/aaqr.2015.05.0376, 2016. a
Bray, C. D., Battye, W., Aneja, V. P., Tong, D. Q., Lee, P., and Tang, Y.: Ammonia emissions from biomass burning in the continental United States, Atmos. Environ., 187, 50–61, https://doi.org/10.1016/j.atmosenv.2018.05.052, 2018. a
Brey, S. J., Ruminski, M., Atwood, S. A., and Fischer, E. V.: Connecting smoke plumes to sources using Hazard Mapping System (HMS) smoke and fire location data over North America, Atmos. Chem. Phys., 18, 1745–1761, https://doi.org/10.5194/acp-18-1745-2018, 2018. a
Burke, M., Childs, M. L., de la Cuesta, B., Qiu, M., Li, J., Gould, C. F., Heft-Neal, S., and Wara, M.: The contribution of wildfire to PM2.5 trends in the USA, Nature, 622, 761–766, https://doi.org/10.1038/s41586-023-06522-6, 2023. a
Carlton, A. G., Turpin, B. J., Altieri, K. E., Seitzinger, S., Reff, A., Lim, H.-J., and Ervens, B.: Atmospheric oxalic acid and SOA production from glyoxal: Results of aqueous photooxidation experiments, Atmos. Environ., 41, 7588–7602, https://doi.org/10.1016/j.atmosenv.2007.05.035, 2007. a, b
Carlton, A. G., Wiedinmyer, C., and Kroll, J. H.: A review of Secondary Organic Aerosol (SOA) formation from isoprene, Atmos. Chem. Phys., 9, 4987–5005, https://doi.org/10.5194/acp-9-4987-2009, 2009. a
Chebbi, A. and Carlier, P.: Carboxylic acids in the troposphere, occurrence, sources, and sinks: A review, Atmos. Environ., 30, 4233–4249, https://doi.org/10.1016/1352-2310(96)00102-1, 1996. a
Chen, G., Canonaco, F., Tobler, A., Aas, W., Alastuey, A., Allan, J., Atabakhsh, S., Aurela, M., Baltensperger, U., Bougiatioti, A., De Brito, J. F., Ceburnis, D., Chazeau, B., Chebaicheb, H., Daellenbach, K. R., Ehn, M., El Haddad, I., Eleftheriadis, K., Favez, O., Flentje, H., Font, A., Fossum, K., Freney, E., Gini, M., Green, D. C., Heikkinen, L., Herrmann, H., Kalogridis, A.-C., Keernik, H., Lhotka, R., Lin, C., Lunder, C., Maasikmets, M., Manousakas, M. I., Marchand, N., Marin, C., Marmureanu, L., Mihalopoulos, N., Močnik, G., Nęcki, J., O'Dowd, C., Ovadnevaite, J., Peter, T., Petit, J.-E., Pikridas, M., Matthew Platt, S., Pokorná, P., Poulain, L., Priestman, M., Riffault, V., Rinaldi, M., Różański, K., Schwarz, J., Sciare, J., Simon, L., Skiba, A., Slowik, J. G., Sosedova, Y., Stavroulas, I., Styszko, K., Teinemaa, E., Timonen, H., Tremper, A., Vasilescu, J., Via, M., Vodička, P., Wiedensohler, A., Zografou, O., Cruz Minguillón, M., and Prévôt, A. S.: European aerosol phenomenology − 8: Harmonised source apportionment of organic aerosol using 22 Year-long ACSM/AMS datasets, Environ. Int., 166, 107325, https://doi.org/10.1016/j.envint.2022.107325, 2022. a
Cong, Z., Kawamura, K., Kang, S., and Fu, P.: Penetration of biomass-burning emissions from South Asia through the Himalayas: new insights from atmospheric organic acids, Sci. Rep.-UK, 5, 9580, https://doi.org/10.1038/srep09580, 2015. a
Cook, R. D., Lin, Y.-H., Peng, Z., Boone, E., Chu, R. K., Dukett, J. E., Gunsch, M. J., Zhang, W., Tolic, N., Laskin, A., and Pratt, K. A.: Biogenic, urban, and wildfire influences on the molecular composition of dissolved organic compounds in cloud water, Atmos. Chem. Phys., 17, 15167–15180, https://doi.org/10.5194/acp-17-15167-2017, 2017. a, b, c
Cooper, O. R., Chang, K., Bates, K., Brown, S. S., Chace, W. S., Coggon, M. M., Gorchov Negron, A. M., Middlebrook, A. M., Peischl, J., Piasecki, A., Schafer, N., Stockwell, C. E., Wang, S., Warneke, C., Zuraski, K., Miyazaki, K., Payne, V. H., Pennington, E. A., Worden, J. R., Bowman, K. W., and McDonald, B. C.: Early Season 2023 Wildfires Generated Record-Breaking Surface Ozone Anomalies Across the U.S. Upper Midwest, Geophys. Res. Lett., 51, https://doi.org/10.1029/2024gl111481, 2024. a
Coury, C. and Dillner, A. M.: A method to quantify organic functional groups and inorganic compounds in ambient aerosols using attenuated total reflectance FTIR spectroscopy and multivariate chemometric techniques, Atmos. Environ., 42, 5923–5932, https://doi.org/10.1016/j.atmosenv.2008.03.026, 2008. a
De Haan, D. O., Hawkins, L. N., Kononenko, J. A., Turley, J. J., Corrigan, A. L., Tolbert, M. A., and Jimenez, J. L.: Formation of Nitrogen-Containing Oligomers by Methylglyoxal and Amines in Simulated Evaporating Cloud Droplets, Environ. Sci. Technol., 45, 984–991, https://doi.org/10.1021/es102933x, 2011. a
De Haan, D. O., Hawkins, L. N., Jansen, K., Welsh, H. G., Pednekar, R., de Loera, A., Jimenez, N. G., Tolbert, M. A., Cazaunau, M., Gratien, A., Bergé, A., Pangui, E., Formenti, P., and Doussin, J.-F.: Glyoxal's impact on dry ammonium salts: fast and reversible surface aerosol browning, Atmos. Chem. Phys., 20, 9581–9590, https://doi.org/10.5194/acp-20-9581-2020, 2020. a
Deguillaume, L., Charbouillot, T., Joly, M., Vaïtilingom, M., Parazols, M., Marinoni, A., Amato, P., Delort, A.-M., Vinatier, V., Flossmann, A., Chaumerliac, N., Pichon, J. M., Houdier, S., Laj, P., Sellegri, K., Colomb, A., Brigante, M., and Mailhot, G.: Classification of clouds sampled at the puy de Dôme (France) based on 10 yr of monitoring of their physicochemical properties, Atmos. Chem. Phys., 14, 1485–1506, https://doi.org/10.5194/acp-14-1485-2014, 2014. a, b
Ditto, J. C., He, M., Hass-Mitchell, T. N., Moussa, S. G., Hayden, K., Li, S.-M., Liggio, J., Leithead, A., Lee, P., Wheeler, M. J., Wentzell, J. J. B., and Gentner, D. R.: Atmospheric evolution of emissions from a boreal forest fire: the formation of highly functionalized oxygen-, nitrogen-, and sulfur-containing organic compounds, Atmos. Chem. Phys., 21, 255–267, https://doi.org/10.5194/acp-21-255-2021, 2021. a, b
Driessen, O. M. and Murphy, J. G.: Assessing pH- and temperature-dependence in the aqueous phase partitioning of organic acids and bases in the atmosphere, Environmental Science: Atmospheres, 5, 591–602, https://doi.org/10.1039/d5ea00034c, 2025. a
Duan, J., Huang, R.-J., Gu, Y., Lin, C., Zhong, H., Wang, Y., Yuan, W., Ni, H., Yang, L., Chen, Y., Worsnop, D. R., and O'Dowd, C.: The formation and evolution of secondary organic aerosol during summer in Xi'an: Aqueous phase processing in fog-rain days, Sci. Total Environ., 756, 144077, https://doi.org/10.1016/j.scitotenv.2020.144077, 2021. a
Dukett, J. E., Aleksic, N., Houck, N., Snyder, P., Casson, P., and Cantwell, M.: Progress toward clean cloud water at Whiteface Mountain New York, Atmos. Environ., 45, 6669–6673, https://doi.org/10.1016/j.atmosenv.2011.08.070, 2011. a
Ervens, B.: Average Cloud Droplet Size and Composition: Good Assumptions for Predicting Oxidants in the Atmospheric Aqueous Phase?, J. Phys. Chem. A, 126, 8295–8304, https://doi.org/10.1021/acs.jpca.2c05527, 2022. a
Ervens, B., Turpin, B. J., and Weber, R. J.: Secondary organic aerosol formation in cloud droplets and aqueous particles (aqSOA): a review of laboratory, field and model studies, Atmos. Chem. Phys., 11, 11069–11102, https://doi.org/10.5194/acp-11-11069-2011, 2011. a, b
Ervens, B., Sorooshian, A., Lim, Y. B., and Turpin, B. J.: Key parameters controlling OH-initiated formation of secondary organic aerosol in the aqueous phase (aqSOA), J. Geophys. Res.-Atmos., 119, 3997–4016, https://doi.org/10.1002/2013JD021021, 2014. a
Eugene, A. J., Pillar-Little, E. A., Colussi, A. J., and Guzman, M. I.: Enhanced Acidity of Acetic and Pyruvic Acids on the Surface of Water, Langmuir, 34, 9307–9313, https://doi.org/10.1021/acs.langmuir.8b01606, 2018. a
Falconer, R. E. and Falconer, P. D.: Determination of cloud water acidity at a mountain observatory in the Adirondack Mountains of New York State, J. Geophys. Res.-Oceans, 85, 7465–7470, https://doi.org/10.1029/JC085iC12p07465, 1980. a
Galloway, J. N., Likens, G. E., Keene, W. C., and Miller, J. M.: The composition of precipitation in remote areas of the world, J. Geophys. Res.-Oceans, 87, 8771–8786, https://doi.org/10.1029/JC087iC11p08771, 1982. a
Gilardoni, S., Massoli, P., Paglione, M., Giulianelli, L., Carbone, C., Rinaldi, M., Decesari, S., Sandrini, S., Costabile, F., Gobbi, G. P., Pietrogrande, M. C., Visentin, M., Scotto, F., Fuzzi, S., and Facchini, M. C.: Direct observation of aqueous secondary organic aerosol from biomass-burning emissions, P. Natl. Acad. Sci. USA, 113, 10013–10018, https://doi.org/10.1073/pnas.1602212113, 2016. a
Gioda, A., Reyes-Rodríguez, G. J., Santos-Figueroa, G., Collett, J. L., Decesari, S., Ramos, M. D. C. K. V., Bezerra Netto, H. J. C., De Aquino Neto, F. R., and Mayol-Bracero, O. L.: Speciation of water-soluble inorganic, organic, and total nitrogen in a background marine environment: Cloud water, rainwater, and aerosol particles, J. Geophys. Res., 116, D05203, https://doi.org/10.1029/2010JD015010, 2011. a
Gong, Q. and Demerjian, K. L.: Measurement and analysis of C2–C10 hydrocarbons at Whiteface Mountain, New York, J. Geophys. Res.-Atmos., 102, 28059–28069, https://doi.org/10.1029/97JD02703, 1997. a, b, c
Green, M. B., Campbell, J. L., Hare, D. K., Higgins, G., Russell, A. G., Solomon, C. T., and Driscoll, C. T.: A Sudden Increase in the Concentration of Dissolved Organic Carbon in Precipitation at a Northeastern US Forest, J. Geophys. Res.-Atmos., 130, e2025JD043488, https://doi.org/10.1029/2025JD043488, 2025. a, b
Grosjean, D., Van Cauwenberghe, K., Schmid, J. P., Kelley, P. E., and Pitts, J. N.: Identification of C3–C10 aliphatic dicarboxylic acids in airborne particulate matter, Environ. Sci. Technol., 12, 313–317, https://doi.org/10.1021/es60139a005, 1978. a
Guo, H., Weber, R. J., and Nenes, A.: High levels of ammonia do not raise fine particle pH sufficiently to yield nitrogen oxide-dominated sulfate production, Sci. Rep.-UK, 7, 12109, https://doi.org/10.1038/s41598-017-11704-0, 2017. a
Hahn, M. W.: Broad diversity of viable bacteria in “sterile” (0.2 μm) filtered water, Res. Microbiol., 155, 688–691, https://doi.org/10.1016/j.resmic.2004.05.003, 2004. a
Hand, J. L., Schichtel, B. A., Malm, W. C., and Pitchford, M. L.: Particulate sulfate ion concentration and SO2 emission trends in the United States from the early 1990s through 2010, Atmos. Chem. Phys., 12, 10353–10365, https://doi.org/10.5194/acp-12-10353-2012, 2012. a
Hatakeyama, S., Ohno, M., Weng, J., Takagi, H., and Akimoto, H.: Mechanism for the formation of gaseous and particulate products from ozone-cycloalkene reactions in air, Environ. Sci. Technol., 21, 52–57, https://doi.org/10.1021/es00155a005, 1987. a
Hawkins, L. N., Lemire, A. N., Galloway, M. M., Corrigan, A. L., Turley, J. J., Espelien, B. M., and De Haan, D. O.: Maillard Chemistry in Clouds and Aqueous Aerosol As a Source of Atmospheric Humic-Like Substances, Environ. Sci. Technol., 50, 7443–7452, https://doi.org/10.1021/acs.est.6b00909, 2016. a
Hayden, K. L., Macdonald, A. M., Gong, W., Toom-Sauntry, D., Anlauf, K. G., Leithead, A., Li, S., Leaitch, W. R., and Noone, K.: Cloud processing of nitrate, J. Geophys. Res.-Atmos., 113, 2007JD009732, https://doi.org/10.1029/2007JD009732, 2008. a
He, Y., Xie, Z., Zhang, H., Liebl, W., Toyama, H., and Chen, F.: Oxidative Fermentation of Acetic Acid Bacteria and Its Products, Front. Microbiol., 13, 879246, https://doi.org/10.3389/fmicb.2022.879246, 2022. a
Hems, R. F., Schnitzler, E. G., Bastawrous, M., Soong, R., Simpson, A. J., and Abbatt, J. P. D.: Aqueous Photoreactions of Wood Smoke Brown Carbon, ACS Earth and Space Chemistry, 4, 1149–1160, https://doi.org/10.1021/acsearthspacechem.0c00117, 2020. a
Hennigan, C. J., Izumi, J., Sullivan, A. P., Weber, R. J., and Nenes, A.: A critical evaluation of proxy methods used to estimate the acidity of atmospheric particles, Atmos. Chem. Phys., 15, 2775–2790, https://doi.org/10.5194/acp-15-2775-2015, 2015. a, b
Herckes, P., Valsaraj, K. T., and Collett, J. L.: A review of observations of organic matter in fogs and clouds: Origin, processing and fate, Atmos. Res., 132–133, 434–449, https://doi.org/10.1016/j.atmosres.2013.06.005, 2013. a
Herrmann, H., Schaefer, T., Tilgner, A., Styler, S. A., Weller, C., Teich, M., and Otto, T.: Tropospheric Aqueous-Phase Chemistry: Kinetics, Mechanisms, and Its Coupling to a Changing Gas Phase, Chem. Rev., 115, 4259–4334, https://doi.org/10.1021/cr500447k, 2015. a
Hilario, M. R. A., Crosbie, E., Bañaga, P. A., Betito, G., Braun, R. A., Cambaliza, M. O., Corral, A. F., Cruz, M. T., Dibb, J. E., Lorenzo, G. R., MacDonald, A. B., Robinson, C. E., Shook, M. A., Simpas, J. B., Stahl, C., Winstead, E., Ziemba, L. D., and Sorooshian, A.: Particulate Oxalate-To-Sulfate Ratio as an Aqueous Processing Marker: Similarity Across Field Campaigns and Limitations, Geophys. Res. Lett., 48, e2021GL096520, https://doi.org/10.1029/2021GL096520, 2021. a
Husain, L., Dutkiewicz, V. A., Hussain, M. M., Khwaja, H. A., Burkhard, E. G., Mehmood, G., Parekh, P. P., and Canelli, E.: A study of heterogeneous oxidation of SO2 in summer clouds, J. Geophys. Res.-Atmos., 96, 18789–18805, https://doi.org/10.1029/91JD01943, 1991. a
Jaakkola, E., Hellén, H., Olin, S., Pleijel, H., Tykkä, T., and Holst, T.: Ozone stress response of leaf BVOC emission and photosynthesis in mountain birch (Betula pubescens spp. czerepanovii) depends on leaf age, Plant-Environment Interactions, 5, e10134, https://doi.org/10.1002/pei3.10134, 2024. a
Jain, P., Barber, Q. E., Taylor, S. W., Whitman, E., Castellanos Acuna, D., Boulanger, Y., Chavardès, R. D., Chen, J., Englefield, P., Flannigan, M., Girardin, M. P., Hanes, C. C., Little, J., Morrison, K., Skakun, R. S., Thompson, D. K., Wang, X., and Parisien, M.-A.: Drivers and Impacts of the Record-Breaking 2023 Wildfire Season in Canada, Nat. Commun., 15, 6764, https://doi.org/10.1038/s41467-024-51154-7, 2024a. a
Jain, P., Sharma, A. R., Acuna, D. C., Abatzoglou, J. T., and Flannigan, M.: Record-breaking fire weather in North America in 2021 was initiated by the Pacific northwest heat dome, Communications Earth & Environment, 5, 202, https://doi.org/10.1038/s43247-024-01346-2, 2024b. a
Jimenez, J. L., Canagaratna, M. R., Donahue, N. M., Prevot, A. S. H., Zhang, Q., Kroll, J. H., DeCarlo, P. F., Allan, J. D., Coe, H., Ng, N. L., Aiken, A. C., Docherty, K. S., Ulbrich, I. M., Grieshop, A. P., Robinson, A. L., Duplissy, J., Smith, J. D., Wilson, K. R., Lanz, V. A., Hueglin, C., Sun, Y. L., Tian, J., Laaksonen, A., Raatikainen, T., Rautiainen, J., Vaattovaara, P., Ehn, M., Kulmala, M., Tomlinson, J. M., Collins, D. R., Cubison, M. J., E., Dunlea, J., Huffman, J. A., Onasch, T. B., Alfarra, M. R., Williams, P. I., Bower, K., Kondo, Y., Schneider, J., Drewnick, F., Borrmann, S., Weimer, S., Demerjian, K., Salcedo, D., Cottrell, L., Griffin, R., Takami, A., Miyoshi, T., Hatakeyama, S., Shimono, A., Sun, J. Y., Zhang, Y. M., Dzepina, K., Kimmel, J. R., Sueper, D., Jayne, J. T., Herndon, S. C., Trimborn, A. M., Williams, L. R., Wood, E. C., Middlebrook, A. M., Kolb, C. E., Baltensperger, U., and Worsnop, D. R.: Evolution of Organic Aerosols in the Atmosphere, Science, 326, 1525–1529, https://doi.org/10.1126/science.1180353, 2009. a
Jin, Y., Luo, H., Tang, S., Xue, S., Nie, C., Peng, X., Zheng, Y., Xu, W., Zhang, G., Pan, X., Sun, Y., Chen, Q., Liu, L., and Zhao, D.: Molecular composition and processing of aqueous secondary organic aerosol in clouds at a mountain site in southeastern China, Atmos. Chem. Phys., 26, 2813–2830, https://doi.org/10.5194/acp-26-2813-2026, 2026. a, b
Jing, B., Peng, C., Wang, Y., Liu, Q., Tong, S., Zhang, Y., and Ge, M.: Hygroscopic properties of potassium chloride and its internal mixtures with organic compounds relevant to biomass burning aerosol particles, Sci. Rep.-UK, 7, 43572, https://doi.org/10.1038/srep43572, 2017. a
Joo, T., Rogers, M. J., Soong, C., Hass-Mitchell, T., Heo, S., Bell, M. L., Ng, N. L., and Gentner, D. R.: Aged and Obscured Wildfire Smoke Associated with Downwind Health Risks, Environ. Sci. Tech. Let., 11, 1340–1347, https://doi.org/10.1021/acs.estlett.4c00785, 2024. a, b
Karl, T., Guenther, A., Spirig, C., Hansel, A., and Fall, R.: Correction to “Seasonal variation of biogenic VOC emissions above a mixed hardwood forest in northern Michigan”, Geophys. Res. Lett., 31, 2003GL019387, https://doi.org/10.1029/2003GL019387, 2004. a
Kawamura, K. and Bikkina, S.: A review of dicarboxylic acids and related compounds in atmospheric aerosols: Molecular distributions, sources and transformation, Atmos. Res., 170, 140–160, https://doi.org/10.1016/j.atmosres.2015.11.018, 2016. a, b
Kawamura, K. and Ikushima, K.: Seasonal changes in the distribution of dicarboxylic acids in the urban atmosphere, Environ. Sci. Technol., 27, 2227–2235, https://doi.org/10.1021/es00047a033, 1993. a
Keene, W. C. and Galloway, J. N.: Considerations regarding sources for formic and acetic acids in the troposphere, J. Geophys. Res.-Atmos., 91, 14466–14474, https://doi.org/10.1029/JD091iD13p14466, 1986. a
Keene, W. C., Galloway, J. N., and Holden, J. D.: Measurement of weak organic acidity in precipitation from remote areas of the world, J. Geophys. Res.-Oceans, 88, 5122–5130, https://doi.org/10.1029/JC088iC09p05122, 1983. a
Khwaja, H. A.: Atmospheric concentrations of carboxylic acids and related compounds at a semiurban site, Atmos. Environ., 29, 127–139, https://doi.org/10.1016/1352-2310(94)00211-3, 1995. a, b
Khwaja, H. A., Brudnoy, S., and Husain, L.: Chemical characterization of three summer cloud episodes at Whiteface Mountain, Chemosphere, 31, 3357–3381, https://doi.org/10.1016/0045-6535(95)00187-D, 1995. a, b, c, d, e, f, g
Khwaja, H. A., Khan, A. R., and Qureshi, S.: Ion Chromatographic Determination of Anions in Environmental Samples, Int. J. Environ. An. Ch., 75, 285–297, https://doi.org/10.1080/03067319908047317, 1999. a
Kim, H., Collier, S., Ge, X., Xu, J., Sun, Y., Jiang, W., Wang, Y., Herckes, P., and Zhang, Q.: Chemical processing of water-soluble species and formation of secondary organic aerosol in fogs, Atmos. Environ., 200, 158–166, https://doi.org/10.1016/j.atmosenv.2018.11.062, 2019. a
Kuang, Y., Luo, B., Huang, S., Liu, J., Hu, W., Peng, Y., Chen, D., Yue, D., Xu, W., Yuan, B., and Shao, M.: Formation of highly absorptive secondary brown carbon through nighttime multiphase chemistry of biomass burning emissions, Atmos. Chem. Phys., 25, 3737–3752, https://doi.org/10.5194/acp-25-3737-2025, 2025. a
Kundu, S., Kawamura, K., Andreae, T. W., Hoffer, A., and Andreae, M. O.: Molecular distributions of dicarboxylic acids, ketocarboxylic acids and α-dicarbonyls in biomass burning aerosols: implications for photochemical production and degradation in smoke layers, Atmos. Chem. Phys., 10, 2209–2225, https://doi.org/10.5194/acp-10-2209-2010, 2010. a
Kunwar, B., Kawamura, K., Fujiwara, S., Fu, P., Miyazaki, Y., and Pokhrel, A.: Dicarboxylic acids, oxocarboxylic acids and α-dicarbonyls in atmospheric aerosols from Mt. Fuji, Japan: Implication for primary emission versus secondary formation, Atmos. Res., 221, 58–71, https://doi.org/10.1016/j.atmosres.2019.01.021, 2019. a
Lance, S.: Whiteface Mountain Cloud Water Chemical Composition 2018–2021, http://atmoschem.asrc.cestm.albany.edu/~cloudwater/pub/Data.htm, last access: 24 January 2023. a
Lance, S., Zhang, J., Schwab, J. J., Casson, P., Brandt, R. E., Fitzjarrald, D. R., Schwab, M. J., Sicker, J., Lu, C.-H., Chen, S.-P., Yun, J., Freedman, J. M., Shrestha, B., Min, Q., Beauharnois, M., Crandall, B., Joseph, E., Brewer, M. J., Minder, J. R., Orlowski, D., Christiansen, A., Carlton, A. G., and Barth, M. C.: Overview of the CPOC Pilot Study at Whiteface Mountain, NY: Cloud Processing of Organics within Clouds (CPOC), B. Am. Meteorol. Soc., 101, E1820–E1841, https://doi.org/10.1175/BAMS-D-19-0022.1, 2020. a, b
Lance, S., Lawrence, C., Tripathy, A., Casson, P., Snyder, P., Murray, G., Murray, D., Wymore, A., McDowell, B., Shattuck, M., Shanley, J., Campbell, J., Green, M., Apel, E., Hornbrook, R., Hills, A., Yerger, E., and Kelting, D.: Long-term Trends in Organic Carbon Concentrations within Cloud Water and Precipitation Samples in the Northeastern United States, EGU General Assembly 2025, Vienna, Austria, 27 Apr–2 May 2025, EGU25-13574, https://doi.org/10.5194/egusphere-egu25-13574, 2025. a
Laskin, A., West, C. P., and Hettiyadura, A. P. S.: Molecular insights into the composition, sources, and aging of atmospheric brown carbon, Chem. Soc. Rev., 54, 1583–1612, https://doi.org/10.1039/D3CS00609C, 2025. a
Lawrence, C., Barth, M., Orlando, J., Casson, P., Brandt, R., Kelting, D., Yerger, E., and Lance, S.: Process analysis of elevated concentrations of organic acids at Whiteface Mountain, New York, Atmos. Chem. Phys., 24, 13693–13713, https://doi.org/10.5194/acp-24-13693-2024, 2024. a, b, c, d
Lawrence, C. E., Casson, P., Brandt, R., Schwab, J. J., Dukett, J. E., Snyder, P., Yerger, E., Kelting, D., VandenBoer, T. C., and Lance, S.: Long-term monitoring of cloud water chemistry at Whiteface Mountain: the emergence of a new chemical regime, Atmos. Chem. Phys., 23, 1619–1639, https://doi.org/10.5194/acp-23-1619-2023, 2023. a, b, c, d, e, f, g, h, i, j, k, l, m, n, o, p, q, r
Lee, A. K. Y., Zhao, R., Li, R., Liggio, J., Li, S.-M., and Abbatt, J. P. D.: Formation of Light Absorbing Organo-Nitrogen Species from Evaporation of Droplets Containing Glyoxal and Ammonium Sulfate, Environ. Sci. Technol., 47, 12819–12826, https://doi.org/10.1021/es402687w, 2013. a
Lee, J. Y., Peterson, P. K., Vear, L. R., Cook, R. D., Sullivan, A. P., Smith, E., Hawkins, L. N., Olson, N. E., Hems, R., Snyder, P. K., and Pratt, K. A.: Wildfire Smoke Influence on Cloud Water Chemical Composition at Whiteface Mountain, New York, J. Geophys. Res.-Atmos., 127, e2022JD037177, https://doi.org/10.1029/2022JD037177, 2022. a, b, c
Lee, T., Yu, X.-Y., Ayres, B., Kreidenweis, S. M., Malm, W. C., and Collett, J. L.: Observations of fine and coarse particle nitrate at several rural locations in the United States, Atmos. Environ., 42, 2720–2732, https://doi.org/10.1016/j.atmosenv.2007.05.016, 2008. a
Li, T., Wang, Z., Wang, Y., Wu, C., Liang, Y., Xia, M., Yu, C., Yun, H., Wang, W., Wang, Y., Guo, J., Herrmann, H., and Wang, T.: Chemical characteristics of cloud water and the impacts on aerosol properties at a subtropical mountain site in Hong Kong SAR, Atmos. Chem. Phys., 20, 391–407, https://doi.org/10.5194/acp-20-391-2020, 2020. a
Lim, Y. B., Tan, Y., Perri, M. J., Seitzinger, S. P., and Turpin, B. J.: Aqueous chemistry and its role in secondary organic aerosol (SOA) formation, Atmos. Chem. Phys., 10, 10521–10539, https://doi.org/10.5194/acp-10-10521-2010, 2010. a
Lim, Y. B., Tan, Y., and Turpin, B. J.: Chemical insights, explicit chemistry, and yields of secondary organic aerosol from OH radical oxidation of methylglyoxal and glyoxal in the aqueous phase, Atmos. Chem. Phys., 13, 8651–8667, https://doi.org/10.5194/acp-13-8651-2013, 2013. a
Liu, J., Zhang, F., Xu, W., Sun, Y., Chen, L., Li, S., Ren, J., Hu, B., Wu, H., and Zhang, R.: Hygroscopicity of Organic Aerosols Linked to Formation Mechanisms, Geophys. Res. Lett., 48, e2020GL091683, https://doi.org/10.1029/2020GL091683, 2021. a
Liu, J. C. and Peng, R. D.: The impact of wildfire smoke on compositions of fine particulate matter by ecoregion in the Western US, J. Expo. Sci. Env. Epid., 29, 765–776, https://doi.org/10.1038/s41370-018-0064-7, 2019. a
Liu, Y., Liggio, J., Staebler, R., and Li, S.-M.: Reactive uptake of ammonia to secondary organic aerosols: kinetics of organonitrogen formation, Atmos. Chem. Phys., 15, 13569–13584, https://doi.org/10.5194/acp-15-13569-2015, 2015. a
Lombardo, S., Tripathy, A., Chittur, S. V., Gentry, D., Hayden, A., Kuentzel, M. L., Casson, P. W., Patel, R., Hammond, L., and Lance, S.: Preliminary study of Microbiology in Clouds at Whiteface Mountain in New York, EGU General Assembly 2025, Vienna, Austria, 27 Apr–2 May 2025, EGU25-7510, https://doi.org/10.5194/egusphere-egu25-7510, 2025. a, b
Lv, S., Wang, F., Wu, C., Chen, Y., Liu, S., Zhang, S., Li, D., Du, W., Zhang, F., Wang, H., Huang, C., Fu, Q., Duan, Y., and Wang, G.: Gas-to-Aerosol Phase Partitioning of Atmospheric Water-Soluble Organic Compounds at a Rural Site in China: An Enhancing Effect of NH3 on SOA Formation, Environ. Sci. Technol., 56, 3915–3924, https://doi.org/10.1021/acs.est.1c06855, 2022. a
Marinoni, A., Laj, P., Sellegri, K., and Mailhot, G.: Cloud chemistry at the Puy de Dôme: variability and relationships with environmental factors, Atmos. Chem. Phys., 4, 715–728, https://doi.org/10.5194/acp-4-715-2004, 2004. a, b
Marracci, M. and Murray, C.: pH-Dependent Kinetics of Imidazole Production in Aqueous Glyoxal/Ammonium Sulfate Microdroplets, ACS Earth and Space Chemistry, 9, 959–969, https://doi.org/10.1021/acsearthspacechem.5c00023, 2025. a
Mattsson, F., Neuberger, A., Heikkinen, L., Gramlich, Y., Paglione, M., Rinaldi, M., Decesari, S., Zieger, P., Riipinen, I., and Mohr, C.: Enrichment of organic nitrogen in fog residuals observed in the Italian Po Valley, Atmos. Chem. Phys., 25, 7973–7989, https://doi.org/10.5194/acp-25-7973-2025, 2025. a
May, W.: atmoschem.process: ASRC Atmospheric Chemistry Data Processing, Zenodo [data set], https://doi.org/10.5281/zenodo.4394567, 2020. a
Michna, P., Werner, R. A., and Eugster, W.: Does fog chemistry in Switzerland change with altitude?, Atmos. Res., 151, 31–44, https://doi.org/10.1016/j.atmosres.2014.02.008, 2015. a
Mochizuki, T., Kawamura, K., Miyazaki, Y., Wada, R., Takahashi, Y., Saigusa, N., and Tani, A.: Secondary formation of oxalic acid and related organic species from biogenic sources in a larch forest at the northern slope of Mt. Fuji, Atmos. Environ., 166, 255–262, https://doi.org/10.1016/j.atmosenv.2017.07.028, 2017. a
Mohnen, V. A.: Cloud water collection from aircraft, Atmos. Technol. (United States), 12, https://www.osti.gov/biblio/6320448 (last access: 17 March 2026), 1979. a
Mohnen, V. A. and Kadlecek, J. A.: Cloud chemistry research at Whiteface Mountain, Tellus B, 41B, 79–91, https://doi.org/10.1111/j.1600-0889.1989.tb00127.x, 1989. a
Mohnen, V. A. and Vong, R. J.: A climatology of cloud chemistry for the eastern United States derived from the mountain cloud chemistry project, Environ. Rev., 1, 38–54, 1993. a
Murray, D. S., Shattuck, M. D., McDowell, W. H., and Wymore, A. S.: Nitrogen wet deposition stoichiometry: the role of organic nitrogen, seasonality, and snow, Biogeochemistry, 160, 301–314, https://doi.org/10.1007/s10533-022-00966-0, 2022. a
Murray, G. L. D., Kimball, K. D., Hill, L. B., Hislop, J. E., and Weathers, K. C.: Long-Term Trends in Cloud and Rain Chemistry on Mount Washington, New Hampshire, Water Air Soil Poll., 224, 1653, https://doi.org/10.1007/s11270-013-1653-7, 2013. a
Myriokefalitakis, S., Tsigaridis, K., Mihalopoulos, N., Sciare, J., Nenes, A., Kawamura, K., Segers, A., and Kanakidou, M.: In-cloud oxalate formation in the global troposphere: a 3-D modeling study, Atmos. Chem. Phys., 11, 5761–5782, https://doi.org/10.5194/acp-11-5761-2011, 2011. a
Ng, N. L., Brown, S. S., Archibald, A. T., Atlas, E., Cohen, R. C., Crowley, J. N., Day, D. A., Donahue, N. M., Fry, J. L., Fuchs, H., Griffin, R. J., Guzman, M. I., Herrmann, H., Hodzic, A., Iinuma, Y., Jimenez, J. L., Kiendler-Scharr, A., Lee, B. H., Luecken, D. J., Mao, J., McLaren, R., Mutzel, A., Osthoff, H. D., Ouyang, B., Picquet-Varrault, B., Platt, U., Pye, H. O. T., Rudich, Y., Schwantes, R. H., Shiraiwa, M., Stutz, J., Thornton, J. A., Tilgner, A., Williams, B. J., and Zaveri, R. A.: Nitrate radicals and biogenic volatile organic compounds: oxidation, mechanisms, and organic aerosol, Atmos. Chem. Phys., 17, 2103–2162, https://doi.org/10.5194/acp-17-2103-2017, 2017. a
Nguyen, T. B., Crounse, J. D., Teng, A. P., Clair, J. M. S., Paulot, F., Wolfe, G. M., and Wennberg, P. O.: Rapid deposition of oxidized biogenic compounds to a temperate forest, P. Natl. Acad. Sci. USA, 112, E392–E401, https://doi.org/10.1073/pnas.1418702112, 2015. a
Nuñez López, L., Amato, P., and Ervens, B.: Bacteria in clouds biodegrade atmospheric formic and acetic acids, Atmos. Chem. Phys., 24, 5181–5198, https://doi.org/10.5194/acp-24-5181-2024, 2024. a
Park, Y. H., Sokolik, I. N., and Hall, S. R.: The Impact of Smoke on the Ultraviolet and Visible Radiative Forcing Under Different Fire Regimes, Air, Soil and Water Research, 11, 1178622118774803, https://doi.org/10.1177/1178622118774803, 2018. a
Paulot, F., Wunch, D., Crounse, J. D., Toon, G. C., Millet, D. B., DeCarlo, P. F., Vigouroux, C., Deutscher, N. M., González Abad, G., Notholt, J., Warneke, T., Hannigan, J. W., Warneke, C., de Gouw, J. A., Dunlea, E. J., De Mazière, M., Griffith, D. W. T., Bernath, P., Jimenez, J. L., and Wennberg, P. O.: Importance of secondary sources in the atmospheric budgets of formic and acetic acids, Atmos. Chem. Phys., 11, 1989–2013, https://doi.org/10.5194/acp-11-1989-2011, 2011. a
Permar, W., Wang, Q., Selimovic, V., Wielgasz, C., Yokelson, R. J., Hornbrook, R. S., Hills, A. J., Apel, E. C., Ku, I., Zhou, Y., Sive, B. C., Sullivan, A. P., Collett, J. L., Campos, T. L., Palm, B. B., Peng, Q., Thornton, J. A., Garofalo, L. A., Farmer, D. K., Kreidenweis, S. M., Levin, E. J. T., DeMott, P. J., Flocke, F., Fischer, E. V., and Hu, L.: Emissions of Trace Organic Gases From Western U.S. Wildfires Based on WE-CAN Aircraft Measurements, J. Geophys. Res.-Atmos., 126, e2020JD033838, https://doi.org/10.1029/2020JD033838, 2021. a
Permar, W., Wielgasz, C., Jin, L., Chen, X., Coggon, M. M., Garofalo, L. A., Gkatzelis, G. I., Ketcherside, D., Millet, D. B., Palm, B. B., Peng, Q., Robinson, M. A., Thornton, J. A., Veres, P., Warneke, C., Yokelson, R. J., Fischer, E. V., and Hu, L.: Assessing formic and acetic acid emissions and chemistry in western U.S. wildfire smoke: implications for atmospheric modeling, Environmental Science: Atmospheres, 3, 1620–1641, https://doi.org/10.1039/d3ea00098b, 2023. a, b
Plessow, K., Acker, K., Heinrichs, H., and Möller, D.: Time study of trace elements and major ions during two cloud events at the Mt. Brocken, Atmos. Environ., 35, 367–378, https://doi.org/10.1016/S1352-2310(00)00134-5, 2001. a
Pye, H. O. T., Nenes, A., Alexander, B., Ault, A. P., Barth, M. C., Clegg, S. L., Collett Jr., J. L., Fahey, K. M., Hennigan, C. J., Herrmann, H., Kanakidou, M., Kelly, J. T., Ku, I.-T., McNeill, V. F., Riemer, N., Schaefer, T., Shi, G., Tilgner, A., Walker, J. T., Wang, T., Weber, R., Xing, J., Zaveri, R. A., and Zuend, A.: The acidity of atmospheric particles and clouds, Atmos. Chem. Phys., 20, 4809–4888, https://doi.org/10.5194/acp-20-4809-2020, 2020. a, b, c
Rattigan, O. V., Dutkiewicz, V. A., Das, M., Judd, C. D., and Husain, L.: Oxidation of SO2 in Clouds at Whiteface Mountain, in: Air-Surface Exchange of Gases and Particles (2000), edited by: Fowler, D., Pitcairn, C., and Erisman, J.-W., Dordrecht, 391–400, https://doi.org/10.1023/A:1013168903087, 2001. a
Roberts, J. M., Veres, P. R., Cochran, A. K., Warneke, C., Burling, I. R., Yokelson, R. J., Lerner, B., Gilman, J. B., Kuster, W. C., Fall, R., and de Gouw, J.: Isocyanic acid in the atmosphere and its possible link to smoke-related health effects, P. Natl. Acad. Sci. USA, 108, 8966–8971, https://doi.org/10.1073/pnas.1103352108, 2011. a
Rogers, M. J., Joo, T., Hass-Mitchell, T., Canagaratna, M. R., Campuzano-Jost, P., Sueper, D., Tran, M. N., Machesky, J. E., Roscioli, J. R., Jimenez, J. L., Krechmer, J. E., Lambe, A. T., Nault, B. A., and Gentner, D. R.: Humid Summers Promote Urban Aqueous-Phase Production of Oxygenated Organic Aerosol in the Northeastern United States, Geophys. Res. Lett., 52, e2024GL112005, https://doi.org/10.1029/2024GL112005, 2025. a
Sagona, J. A., Dukett, J. E., Hawley, H. A., and Mazurek, M. A.: Sequential derivatization of polar organic compounds in cloud water using O-(2,3,4,5,6-pentafluorobenzyl)hydroxylamine hydrochloride, N,O-bis(trimethylsilyl)trifluoroacetamide, and gas-chromatography/mass spectrometry analysis, J. Chromatogr. A, 1362, 16–24, https://doi.org/10.1016/j.chroma.2014.08.001, 2014. a
Salma, I., Ocskay, R., and Láng, G. G.: Properties of atmospheric humic-like substances – water system, Atmos. Chem. Phys., 8, 2243–2254, https://doi.org/10.5194/acp-8-2243-2008, 2008. a
Schwab, J. J., Wolfe, D., Casson, P., Brandt, R., Demerjian, K. L., Husain, L., Dutkiewicz, V. A., Civerolo, K. L., and Rattigan, O. V.: Atmospheric Science Research at Whiteface Mountain, NY: Site Description and History, Aerosol Air Qual. Res., 16, 827–840, https://doi.org/10.4209/aaqr.2015.05.0343, 2016. a, b
Seinfeld, J. H. and Pandis, S. N.: Atmospheric Chemistry and Physics: From Air Pollution to Climate Change, 3rd edn., John Wiley & Sons, Hoboken, NJ, ISBN 978-1-119-22117-3, 2016. a
Shah, V., Jacob, D. J., Moch, J. M., Wang, X., and Zhai, S.: Global modeling of cloud water acidity, precipitation acidity, and acid inputs to ecosystems, Atmos. Chem. Phys., 20, 12223–12245, https://doi.org/10.5194/acp-20-12223-2020, 2020. a
Shen, M., Li, J., Liu, Y., Dai, W., Wang, G., Qi, W., Chen, Y., Guo, X., Zhang, Y., Li, L., Cao, Y., Feng, Q., Su, H., and Cao, J.: Comparison of acidity and chemical composition of summertime cloud water and aerosol at an alpine site in Northwest China: Implications for the neutral property of clouds in the free troposphere, Sci. Total Environ., 925, 171775, https://doi.org/10.1016/j.scitotenv.2024.171775, 2024. a
Shrestha, B., Brotzge, J. A., and Wang, J.: Observations and Impacts of Long-Range Transported Wildfire Smoke on Air Quality Across New York State During July 2021, Geophys. Res. Lett., 49, https://doi.org/10.1029/2022gl100216, 2022. a
Shu, J., Zhang, R., Abudumutailifu, M., Cai, D., Liu, S., Li, C., Pan, X., Liu, L., Mellouki, A., and Chen, J.-M.: Diverse Nitrogen-Containing Organic Compounds in Cloud Water Formed via Nucleophilic Addition Reaction Between Carboxylic Acid and Ammonia, J. Geophys. Res.-Atmos., 130, https://doi.org/10.1029/2025JD043913, 2025. a, b
Slade, J. H., Ault, A. P., Bui, A. T., Ditto, J. C., Lei, Z., Bondy, A. L., Olson, N. E., Cook, R. D., Desrochers, S. J., Harvey, R. M., Erickson, M. H., Wallace, H. W., Alvarez, S. L., Flynn, J. H., Boor, B. E., Petrucci, G. A., Gentner, D. R., Griffin, R. J., and Shepson, P. B.: Bouncier Particles at Night: Biogenic Secondary Organic Aerosol Chemistry and Sulfate Drive Diel Variations in the Aerosol Phase in a Mixed Forest, Environ. Sci. Technol., 53, 4977–4987, https://doi.org/10.1021/acs.est.8b07319, 2019. a
Smolyakov, B. S., Makarov, V. I., Shinkorenko, M. P., Popova, S. A., and Bizin, M. A.: Effects of Siberian wildfires on the chemical composition and acidity of atmospheric aerosols of remote urban, rural and background territories, Environ. Pollut., 188, 8–16, https://doi.org/10.1016/j.envpol.2014.01.017, 2014. a
Sorooshian, A., Varutbangkul, V., Brechtel, F. J., Ervens, B., Feingold, G., Bahreini, R., Murphy, S. M., Holloway, J. S., Atlas, E. L., Buzorius, G., Jonsson, H., Flagan, R. C., and Seinfeld, J. H.: Oxalic acid in clear and cloudy atmospheres: Analysis of data from International Consortium for Atmospheric Research on Transport and Transformation 2004, J. Geophys. Res.-Atmos., 111, 2005JD006880, https://doi.org/10.1029/2005JD006880, 2006. a
Sullivan, A. P., Pokhrel, R. P., Shen, Y., Murphy, S. M., Toohey, D. W., Campos, T., Lindaas, J., Fischer, E. V., and Collett Jr., J. L.: Examination of brown carbon absorption from wildfires in the western US during the WE-CAN study, Atmos. Chem. Phys., 22, 13389–13406, https://doi.org/10.5194/acp-22-13389-2022, 2022. a
Sun, L., Wang, Y., Yue, T., Yang, X., Xue, L., and Wang, W.: Evaluation of the behavior of clouds in a region of severe acid rain pollution in southern China: species, complexes, and variations, Environ. Sci. Pollut. R., 22, 14280–14290, https://doi.org/10.1007/s11356-015-4674-5, 2015. a
Tan, Y., Carlton, A. G., Seitzinger, S. P., and Turpin, B. J.: SOA from methylglyoxal in clouds and wet aerosols: Measurement and prediction of key products, Atmos. Environ., 44, 5218–5226, https://doi.org/10.1016/j.atmosenv.2010.08.045, 2010. a
Tao, Y. and Murphy, J. G.: The sensitivity of PM2.5 acidity to meteorological parameters and chemical composition changes: 10 year records from six Canadian monitoring sites, Atmos. Chem. Phys., 19, 9309–9320, https://doi.org/10.5194/acp-19-9309-2019, 2019. a, b
Tilgner, A., Schaefer, T., Alexander, B., Barth, M., Collett Jr., J. L., Fahey, K. M., Nenes, A., Pye, H. O. T., Herrmann, H., and McNeill, V. F.: Acidity and the multiphase chemistry of atmospheric aqueous particles and clouds, Atmos. Chem. Phys., 21, 13483–13536, https://doi.org/10.5194/acp-21-13483-2021, 2021. a
Tripathy, A., Khwaja, H., Hussain, M., and Lance, S.: Determination of Low Molecular Weight Organic Acids in Environmental Samples by Ion Chromatography Method, https://doi.org/10.31223/X50449, 2024. a
VandenBoer, T. C., Petroff, A., Markovic, M. Z., and Murphy, J. G.: Size distribution of alkyl amines in continental particulate matter and their online detection in the gas and particle phase, Atmos. Chem. Phys., 11, 4319–4332, https://doi.org/10.5194/acp-11-4319-2011, 2011. a, b
van Pinxteren, D., Fomba, K. W., Mertes, S., Müller, K., Spindler, G., Schneider, J., Lee, T., Collett, J. L., and Herrmann, H.: Cloud water composition during HCCT-2010: Scavenging efficiencies, solute concentrations, and droplet size dependence of inorganic ions and dissolved organic carbon, Atmos. Chem. Phys., 16, 3185–3205, https://doi.org/10.5194/acp-16-3185-2016, 2016. a, b, c
Wang, Y., Guo, J., Wang, T., Ding, A., Gao, J., Zhou, Y., Collett, J. L., and Wang, W.: Influence of regional pollution and sandstorms on the chemical composition of cloud/fog at the summit of Mt. Taishan in northern China, Atmos. Res., 99, 434–442, https://doi.org/10.1016/j.atmosres.2010.11.010, 2011. a
Wang, Z., Wang, Z., Zou, Z., Chen, X., Wu, H., Wang, W., Su, H., Li, F., Xu, W., Liu, Z., and Zhu, J.: Severe Global Environmental Issues Caused by Canada’s Record-Breaking Wildfires in 2023, Adv. Atmos. Sci., 41, 565–571, https://doi.org/10.1007/s00376-023-3241-0, 2024. a
Xu, Y., Miyazaki, Y., Tachibana, E., Sato, K., Ramasamy, S., Mochizuki, T., Sadanaga, Y., Nakashima, Y., Sakamoto, Y., Matsuda, K., and Kajii, Y.: Aerosol Liquid Water Promotes the Formation of Water-Soluble Organic Nitrogen in Submicrometer Aerosols in a Suburban Forest, Environ. Sci. Technol., 54, 1406–1414, https://doi.org/10.1021/acs.est.9b05849, 2020. a
Yang, L., Ray, M. B., and Yu, L. E.: Photooxidation of dicarboxylic acids – Part II: Kinetics, intermediates and field observations, Atmos. Environ., 42, 868–880, https://doi.org/10.1016/j.atmosenv.2007.10.030, 2008. a
Yu, F., Nair, A. A., and Luo, G.: Long-Term Trend of Gaseous Ammonia Over the United States: Modeling and Comparison With Observations, J. Geophys. Res.-Atmos., 123, 8315–8325, https://doi.org/10.1029/2018JD028412, 2018. a
Zhang, J., Lance, S., Brandt, R., Marto, J., Ninneman, M., and Schwab, J.: Observed below-Cloud and Cloud Interstitial Submicron Aerosol Chemical and Physical Properties at Whiteface Mountain, New York, during August 2017, ACS Earth and Space Chemistry, 3, 1438–1450, https://doi.org/10.1021/acsearthspacechem.9b00117, 2019. a
Zhao, W., Wang, Z., Li, S., Li, L., Wei, L., Xie, Q., Yue, S., Li, T., Liang, Y., Sun, Y., Wang, Z., Li, X., Kawamura, K., Wang, T., and Fu, P.: Water-soluble low molecular weight organics in cloud water at Mt. Tai Mo Shan, Hong Kong, Sci. Total Environ., 697, 134095, https://doi.org/10.1016/j.scitotenv.2019.134095, 2019. a
Zhao, Y. and Gao, Y.: Mass size distributions of water-soluble inorganic and organic ions in size-segregated aerosols over metropolitan Newark in the US east coast, Atmos. Environ., 42, 4063–4078, https://doi.org/10.1016/j.atmosenv.2008.01.032, 2008. a
Zheng, B., Ciais, P., Chevallier, F., Yang, H., Canadell, J. G., Chen, Y., van der Velde, I. R., Aben, I., Chuvieco, E., Davis, S. J., Deeter, M., Hong, C., Kong, Y., Li, H., Li, H., Lin, X., He, K., and Zhang, Q.: Record-high CO2 emissions from boreal fires in 2021, Science, 379, 912–917, https://doi.org/10.1126/science.ade0805, 2023a. a
Zheng, G., Su, H., and Cheng, Y.: Role of Carbon Dioxide, Ammonia, and Organic Acids in Buffering Atmospheric Acidity: The Distinct Contribution in Clouds and Aerosols, Environ. Sci. Technol., 57, 12571–12582, https://doi.org/10.1021/acs.est.2c09851, 2023b. a, b
Zuo, Y. and Zhan, J.: Effects of oxalate on Fe-catalyzed photooxidation of dissolved sulfur dioxide in atmospheric water, Atmos. Environ., 39, 27–37, https://doi.org/10.1016/j.atmosenv.2004.09.058, 2005. a




